Medtronic plc has officially launched its MiniMed 780G system in the United States with integrated support for the Instinct sensor, a continuous glucose monitoring (CGM) device developed by Abbott. This broad commercial release follows U.S. Food and Drug Administration clearance earlier in 2025 and represents a pivotal moment for automated insulin delivery systems that combine CGM accuracy with algorithmic dosing precision.
The Instinct sensor, which features up to 15-day wear and a discreet design, is exclusive to Medtronic systems and is now shipping nationwide. While the launch enhances Medtronic’s diabetes portfolio, the real story lies in how this partnership could reshape competitive dynamics across the CGM and insulin delivery markets—and potentially restore Medtronic’s reputation as a serious contender in the closed-loop diabetes care space.
What this launch reveals about Medtronic’s pivot from full-stack to flexible CGM integration
For years, Medtronic has pursued a vertically integrated approach, pairing its insulin pumps with in-house Guardian CGMs. But persistent criticism around Guardian sensor accuracy, comfort, and lag time has forced a strategic rethink. The decision to adopt Abbott’s Instinct CGM for integration into its MiniMed ecosystem signals a new modular mindset—one that prioritizes patient usability and optionality over control of the entire product stack.
With this launch, Medtronic now offers a tiered sensor strategy that includes the Guardian 4, the Simplera Sync, and now the Instinct sensor. Each of these targets a different segment of users based on wear time, form factor, and data features. That diversification may help Medtronic reduce churn, enhance algorithm performance through sensor-driven improvements, and compete more effectively with hybrid-loop platforms powered by Dexcom and used by Tandem Diabetes Care and Insulet Corporation.
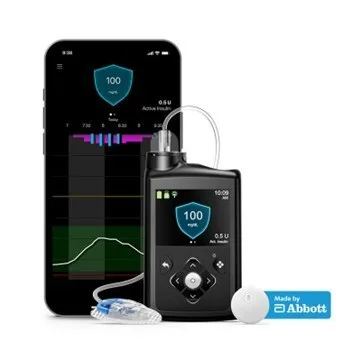
Clinicians tracking the AID space have long pointed to patient dissatisfaction with Guardian CGM wearability and complexity. The Instinct sensor, in contrast, brings Medtronic closer to parity with rivals offering more discreet, longer-wear sensors with minimal calibration requirements. This could significantly strengthen patient retention and onboarding in a market where switching platforms can carry both clinical and financial friction.
Why Abbott’s Instinct sensor could be a quiet disruptor in integrated diabetes systems
Abbott Laboratories has historically focused its CGM business on the FreeStyle Libre franchise, which dominates the global standalone CGM market. Its move into exclusive partnerships for integrated systems marks a strategic broadening of scope. The Instinct sensor, while sharing some design DNA with Libre 3, is uniquely tailored for automated insulin delivery and brings wearability features—like being the smallest and thinnest sensor on the market—to a space that has often lagged in user-centric design.
For Abbott, this partnership with Medtronic is not just about diversifying product applications. It is about establishing a foothold in the integrated CGM market where Dexcom has enjoyed first-mover advantage. Unlike Libre sensors, which have struggled with U.S. AID compatibility, Instinct was built for this specific use case, making it Abbott’s most direct shot at penetrating insulin pump ecosystems.
Industry observers suggest this integration may also help Abbott hedge against pricing pressure in the standalone CGM market, particularly as more payers demand tighter alignment between CGM usage and clinical actionability.
What the MiniMed 780G with Instinct means for closed-loop system innovation
The MiniMed 780G system has already earned a reputation for one of the most aggressive insulin automation algorithms currently available in commercial AID systems, with adjustments every five minutes and automated correction boluses. When paired with the Instinct sensor’s real-time glucose readings and 15-day wear cycle, the system aims to lower patient burden while increasing time in range (TIR)—a primary clinical outcome for people living with type 1 diabetes.
Patients and physicians in early access programs have reported improved day-to-day management, particularly in terms of fewer fingersticks, fewer alarms, and reduced glycemic variability. These experiential improvements may sound incremental, but they target key barriers to CGM and pump adoption: friction in device use and alarm fatigue.
However, long-term, large-cohort data from real-world deployments remain limited, and payer coverage for new sensor configurations often trails clinical adoption. As such, the impact of this pairing on overall health economics, especially in pediatric and underserved populations, will be closely watched.
Why regulators and payers will be paying close attention to this integration model
The U.S. Food and Drug Administration’s clearance of the Instinct sensor specifically for use with the MiniMed 780G underscores a growing regulatory focus on validated sensor-algorithm pairings. In contrast to previous systems where sensors and pumps were sometimes treated as modular plug-ins, new integrated systems are expected to undergo specific performance testing as a combined unit.
This regulatory precedent could influence how other companies approach their own CGM-insulin delivery combinations. There may be increased scrutiny on not just the standalone accuracy or safety of sensors, but also how they perform when coupled with algorithm-driven insulin dosing platforms. It also raises questions about future interoperability. Will CGMs like Instinct be allowed on other AID systems, or will proprietary lock-in remain the norm?
Payers, meanwhile, will want to see data that shows a reduction in emergency events, hospitalizations, or long-term complications when this system is used consistently. Without reimbursement alignment, even the most technically advanced systems can face slow uptake.
What adoption and scalability challenges still stand in the way
Despite the promise of this pairing, Medtronic and Abbott face logistical and commercial hurdles. Scaling sensor manufacturing to support consistent 15-day wear cycles with minimal defects will be critical, especially given the higher expectations placed on integrated CGM systems. Unlike standalone CGMs, integrated systems require ultra-consistent performance to avoid dosing errors.
There is also the matter of patient education. With three different sensors now in play—Guardian 4, Simplera Sync, and Instinct—Medtronic must invest in clinician and patient onboarding to ensure proper matching and expectations. Misalignment between user needs and sensor capabilities could risk negative sentiment, which has historically been a weak spot for Medtronic’s diabetes division.
Finally, questions remain about international rollout, pediatric approval timelines, and whether Abbott will deepen or diversify its integration partnerships in the future. These will all shape how far and fast this technology diffuses across markets.
Why this launch could reset the CGM-insulin ecosystem over the next 12 months
Taken together, the MiniMed 780G system integrated with the Instinct sensor represents more than a product release. It signals a broader shift in strategy for both Medtronic and Abbott, away from single-vendor verticalism and toward ecosystem optimization based on usability and precision.
The CGM-insulin pump space is evolving from closed silos to data- and design-driven platforms. Medtronic’s willingness to adopt external sensor technology suggests that clinical outcomes and patient preferences are starting to overrule legacy control models. Abbott, meanwhile, is staking a new claim in a territory where it has long been absent: integrated insulin delivery.
Over the next year, the ability of this pairing to deliver consistent glycemic outcomes, simplify patient experience, and meet regulatory expectations will determine whether it becomes a case study in successful CGM-insulin convergence—or just another niche launch in a crowded device landscape.