Can Convergent Therapeutics define the next prostate cancer standard after Lu-PSMA therapy?
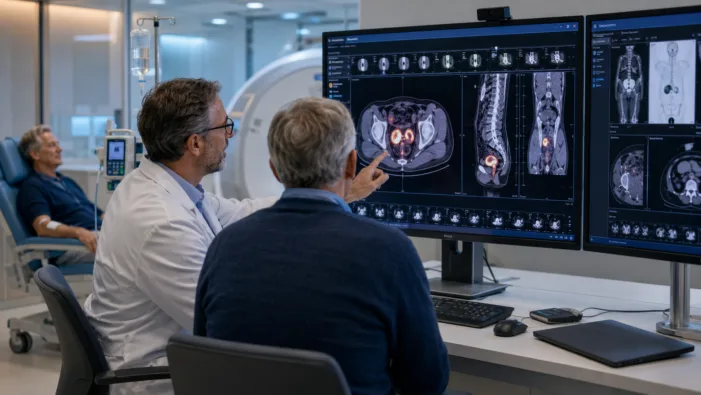
CONV01-α is targeting the post-Lu-PSMA prostate cancer gap. Convergent now faces the Phase 3 test that will decide its real value.

CONV01-α is targeting the post-Lu-PSMA prostate cancer gap. Convergent now faces the Phase 3 test that will decide its real value.

T-CURX and Pantherna are combining CAR-T, mRNA and LNP platforms. Europe’s in vivo cell therapy race just became more serious.
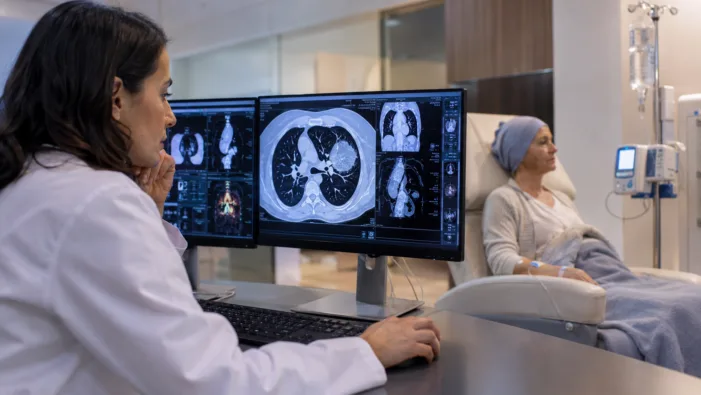
Amgen’s Imdylltra has European approval in relapsed small cell lung cancer. Survival data now face reimbursement and adoption tests.
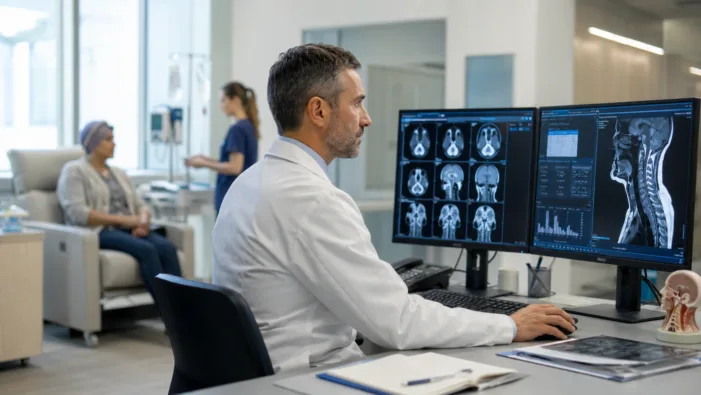
Fulgent Genetics has a diagnostics base and a cancer drug signal. FID-007 now faces the tougher Phase 3 test.

Minghui and Qilu’s MHB088C data show durable disease control in mCRPC. Find out why the B7-H3 ADC still faces key clinical tests.
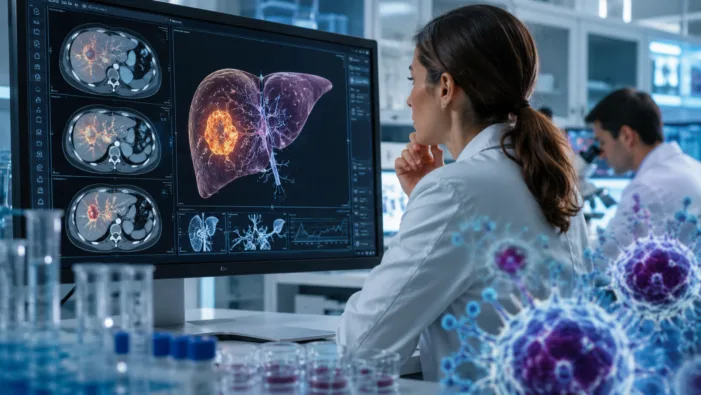
Find out how Oricell’s Ori-C101 CAR-T data could reshape expectations for late-line liver cancer treatment after ASCO 2026.

Read why Lilly’s Hanmi sonefpeglutide deal could reshape long-acting GLP-2 therapy for short bowel syndrome.

Genentech advances enicepatide and petrelintide obesity data at ADA 2026. Find out why Roche’s strategy matters now.
Ivonescimab showed an overall survival benefit in HARMONi-6. Read how the data could reshape first-line squamous NSCLC treatment.

Pfizer’s Braftovi data strengthen its role in BRAF V600E metastatic colorectal cancer. Find out what this could change next.