RevBio Inc. has received clearance from the United States Food and Drug Administration to begin a pilot clinical trial of TETRANITE, its regenerative bone adhesive, for dental ridge augmentation. The study will assess the product’s ability to support bone grafting procedures without the use of membranes, meshes, tacks, or screws—offering a potential simplification to a traditionally complex and failure-prone part of dental implant workflows.
Why TETRANITE’s approach could redefine ridge augmentation procedures
RevBio’s core thesis hinges on the ability of TETRANITE to function as both scaffold and sealant, adhering directly to bony walls while resisting soft tissue ingrowth—one of the major causes of graft failure. Unlike particulate bone graft materials that require secondary fixation and containment, this bioengineered platform claims to offer an all-in-one structure that maintains volume and mechanical integrity during early healing phases.
In dental implantology, especially for patients with significant alveolar bone loss, ridge augmentation is often a prerequisite. The procedure not only adds time and complexity to treatment but also introduces failure risk when grafts shift, resorb prematurely, or get contaminated by soft tissue. According to clinical estimates cited in the announcement, over 30 percent of current bone graft procedures fail to deliver the desired results, prompting costly and time-intensive repeat interventions.
TETRANITE’s ability to maintain structural volume and support natural bone substitution during healing presents a promising counter to this recurring challenge. If clinical outcomes match preclinical claims, this could reduce patient morbidity, cut treatment cycles by months, and lower the financial burden on both providers and payers.
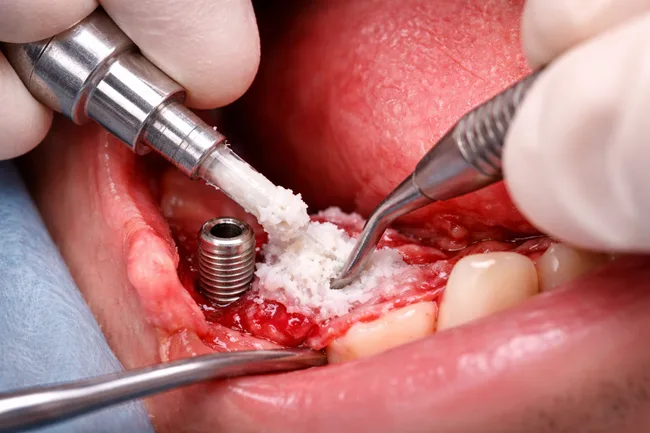
What this reveals about RevBio’s broader platform strategy
While the dental application marks a notable expansion, RevBio is clearly positioning TETRANITE as a multi-specialty bone adhesive with applicability in neurosurgical and orthopedic indications as well. The platform is already involved in 10 separate FDA-approved clinical trials across multiple verticals. Although none of these use cases have yet reached commercial stage, the company is advancing toward De Novo classification pathways that could expedite first market entry.
The TETRANITE platform stands out in part due to its wet-field adhesion—a key hurdle in bone repair technologies, especially in cranial or spinal surgeries where blood and fluid exposure impedes traditional cement or scaffold performance. In that context, the ridge augmentation trial acts not only as a proof point for dental workflows, but as a validation of the platform’s core material science under complex anatomical and procedural conditions.
Analysts tracking the regenerative biomaterials space will likely view this trial as a low-risk, high-visibility milestone with broader implications for RevBio’s value proposition in orthobiologics.
What the FDA greenlight changes for future commercial pathways
FDA’s approval to proceed with a first-in-human study specifically for dental ridge augmentation moves RevBio closer to defining a reimbursable, specialty-specific use case for TETRANITE. Regulatory watchers believe that such indication-specific trials will be critical to De Novo classification success, especially given the broad potential applications of the technology.
By isolating the dental ridge augmentation procedure as a standalone target, RevBio reduces the regulatory ambiguity associated with platform technologies. This segmented strategy can help the company generate targeted clinical evidence for each pathway, bolstering both payer confidence and procedural adoption by clinicians.
Moreover, industry observers suggest that the approval also signals FDA’s increasing openness to biomimetic adhesives and alternative scaffold materials that depart from particulate and membrane-based paradigms. If TETRANITE successfully meets safety and efficacy thresholds in this trial, similar designations for craniofacial or orthopedic augmentation procedures may become easier to navigate.
What risks and uncertainties still remain
Despite the momentum, several risks persist. The transition from preclinical performance to real-world surgical outcomes is often marked by variability in biomaterial integration, especially in patients with comorbidities or compromised bone metabolism. Clinical success will hinge on how well TETRANITE adapts to operator technique, defect morphology, and patient-specific healing dynamics.
Another open question lies in surgeon adoption. Ridge augmentation procedures vary widely across geographies and provider types, with many oral surgeons deeply entrenched in using membranes, meshes, or titanium screws as standard protocol. Shifting clinical practice patterns requires not just better performance, but also favorable reimbursement codes, procedural training, and long-term follow-up data.
Additionally, RevBio’s current positioning as a clinical-stage company without an approved commercial product means its capitalization strategy remains linked to grant funding and early investor confidence. The $1.8 million in Translational Resource Center grants and $2 million from a Small Business Innovation Research (SBIR) program have carried the dental adhesive this far, but commercialization will demand scaled manufacturing and payer engagement, which remain unresolved at this stage.
What the industry will be watching as the trial progresses
Clinicians and industry analysts alike will track several key variables during the pilot trial. These include the rate of bone regeneration, implant volume preservation, complication rates (particularly infection or graft failure), and time to implant readiness. More broadly, the trial will serve as a bellwether for how well multifunctional biomaterials like TETRANITE can compress procedural timelines while maintaining safety.
Success in this trial could open the door for RevBio to push toward a modular product portfolio—customized for dental, neurosurgical, or orthopedic markets—with tailored procedural kits, dosing formats, and delivery instruments. The scalability of such a platform, however, will depend not only on material science but also on integration into existing surgical workflows and provider economics.
In sum, RevBio’s FDA-approved clinical study for dental ridge augmentation represents more than a technical milestone. It serves as a pivotal moment in the validation arc of TETRANITE as a next-generation bone adhesive, and potentially a shift away from the fragmented and failure-prone components that define current bone grafting protocols.
What are the key takeaways from RevBio’s FDA-cleared dental trial for TETRANITE?
- RevBio Inc. has received FDA approval to begin a pilot trial of TETRANITE for dental ridge augmentation.
- The trial aims to eliminate the need for membranes, meshes, or screws in bone graft procedures.
- TETRANITE adheres to bone and resists soft tissue ingrowth while maintaining structural volume.
- Over 30 percent of current ridge augmentations fail, often requiring regrafting.
- This indication-specific trial could de-risk the platform’s path to broader regulatory approvals.
- RevBio is seeking De Novo classification for the TETRANITE platform across multiple specialties.
- Grant funding from the National Institute of Dental and Cranial Research has supported clinical development.
- Surgeons, payers, and regulators will be watching real-world performance, especially around procedural efficiency and outcomes.