The recent U.S. Food and Drug Administration (FDA) approval for RevBio Inc. to begin a pilot trial of its regenerative bone adhesive, TETRANITE, in dental ridge augmentation has reignited industry attention around a long-elusive biomaterials category: viable, clinically integrated bone adhesives. While dental applications are the entry point, the real strategic question is whether these new-generation biomimetic glues are finally poised to replace legacy materials such as polymethyl methacrylate (PMMA) and brittle ceramic scaffolds across orthopedic and cranial repair.
The answer may depend not just on adhesion strength or osteoconductivity, but on whether these materials can withstand the procedural, regulatory, and reimbursement scrutiny that has held this space back for decades.
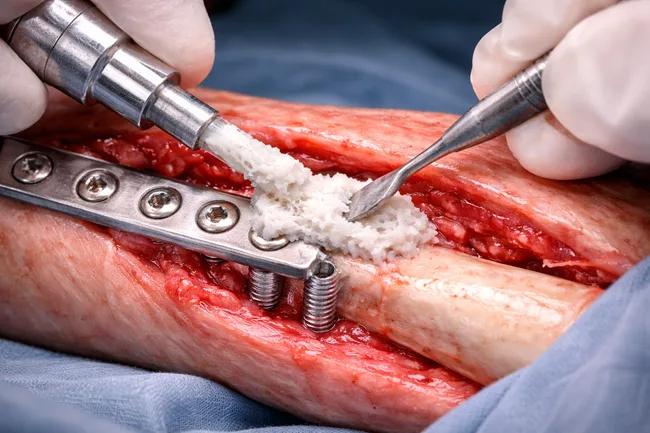
Why bone adhesives failed to gain traction despite decades of innovation attempts
The concept of using glue to bind bone segments or anchor implants has existed for over 50 years. However, the practical execution of this concept has been riddled with problems. Historically, early bone adhesives either lacked sufficient mechanical strength, triggered immune responses, degraded unpredictably, or failed to support the healing cascade required for full osseointegration.
Most available materials in clinical use today still follow the two-component model: a structural filler (such as ceramic, allograft, or calcium phosphate) and a containment mechanism (like screws, plates, or meshes). Attempts to collapse these into a single adhesive-based construct have mostly failed in human use. The rare exceptions—such as fibrin glues—are limited to soft tissue sealing, not structural bone bonding.
Clinicians tracking the category often point to a persistent “functionality gap.” Either the material binds well but lacks biological integration, or it integrates well but lacks strength. Bridging that tradeoff has been the central challenge of this category.
What makes PMMA cement durable but clinically restrictive in modern orthopedic surgery
Polymethyl methacrylate, or PMMA, is still the most widely used material for vertebroplasty, joint anchoring, and temporary fixation in orthopedic settings. It offers rapid curing, predictable setting, and excellent compressive strength. It is, however, completely bioinert. PMMA does not integrate with bone, and over time can lead to implant loosening, micromotion, or mechanical fatigue at the interface.
Its exothermic curing process can cause thermal damage to surrounding tissues, and its lack of resorption or remodeling makes it unsuitable for pediatric, craniofacial, or trauma cases requiring dynamic bone growth. Moreover, PMMA’s inability to function in wet surgical fields without strict preparation has made it suboptimal for use in minimally invasive or emergency procedures.
The result is a material that, while still dominant in operating rooms, is now viewed by many clinicians as a compromise product—useful for immediate structural stabilization, but far from ideal for long-term outcomes or regenerative integration.
How biomimetic bone adhesives aim to replace both fixation hardware and scaffold systems
What sets the next generation of bone adhesives apart is their ambition to serve as both fixation and scaffold. These materials are not just glues in the traditional sense. They are engineered matrices that exhibit surface adhesion, wet-field operability, mechanical strength, and osteoconductivity in a single formulation.
RevBio’s TETRANITE, for example, is designed to bond directly to bone surfaces, resist soft tissue ingress, and eventually be replaced by native bone over time. It is currently under trial in dental augmentation, but the platform is being developed for spinal, cranial, and trauma use cases as well. The company claims its biomaterial performs well in wet fields, maintains mechanical integrity for weeks, and is fully resorbed over a clinically relevant healing timeline.
Similarly, OsStic, a product developed by a UK-Australia consortium, is based on a phosphoserine-calcium compound that acts as a bioglue capable of withstanding compressive and shear forces. Early-stage studies suggest that it can fix small bone fragments without screws or plates, with high initial fixation and subsequent osseointegration.
These materials blur the line between cement, graft, and barrier. Rather than using screws to hold grafts in place while a scaffold promotes healing, these adhesives offer the possibility of a single application event that sets quickly, stays in place, and actively participates in bone regeneration.
Why TETRANITE and OsStic are attracting clinical and regulatory attention
The convergence of regulatory openness, clinical demand, and platform engineering is now giving these materials a second look. TETRANITE’s FDA-approved trial in humans marks a departure from the long preclinical phase that most adhesive candidates never graduate from. Its synthetic composition avoids many of the immunological concerns associated with collagen-based or xenograft products. The support from the National Institute of Dental and Cranial Research, through both TRC grants and SBIR funding, highlights the strategic intent to bring high-impact biomaterials closer to clinical translation.
Meanwhile, OsStic is preparing for broader orthopedic use cases, including craniofacial fracture fixation and osteotomy applications. The material’s dry storage format and room-temperature stability are appealing in both surgical and field hospital environments. Clinical adoption will still depend on whether it can meet regulatory requirements across various anatomical regions and show safety in load-bearing scenarios.
While both platforms are still in early trial phases, their trajectory suggests a growing institutional willingness to validate bone adhesives not just as adjuncts, but as procedural replacements for hardware-intensive interventions.
What barriers still stand in the way of widespread clinical adoption
Despite the renewed interest, several challenges remain. The first is scalability. Manufacturing adhesive biomaterials with consistent performance characteristics, especially at commercial scale, requires tight control of formulation chemistry, storage conditions, and delivery systems.
Second, these materials must perform in a wide range of anatomical environments, from small cranial segments to larger load-bearing structures in the femur or spine. The biomechanical demands vary greatly, and no single formulation is likely to meet all use cases. Tailored product families may be required, each with different curing profiles, strength thresholds, and resorption timelines.
Third, clinicians will need training to apply these materials correctly. Adhesive behavior in live bone, especially in cases with poor vascularization or infection risk, is difficult to predict. The learning curve may slow adoption in non-specialist settings unless accompanied by robust educational programs.
Fourth, regulatory clarity is still emerging. The U.S. Food and Drug Administration has yet to approve a bone adhesive product for a full range of orthopedic indications. Current approvals are limited to dental and cranial uses under tightly defined protocols. Broadening that scope will require not just successful trials, but also robust post-market surveillance data.
Finally, payer reimbursement models must evolve. Today’s coding and billing frameworks are not designed to accommodate hybrid products that function as both scaffold and fixation device. This misalignment may delay hospital adoption, even if clinical interest is strong.
Why procedural convergence may matter more than graft volume in future implant workflows
The potential of bone adhesives like TETRANITE and OsStic lies not in matching or exceeding graft volume statistics alone, but in streamlining how surgeons approach structural reconstruction. In an era of minimally invasive surgery, bundled payments, and outpatient implant workflows, the ability to fix, fill, and regenerate with one material could drive profound shifts in surgical protocol.
The next generation of adhesives is emerging not just as a replacement for PMMA or a supplement to grafts, but as an integrated solution that collapses hardware, biologics, and barriers into a single platform. If validated across broader use cases, these materials could make traditional plates and screws less necessary in small-fracture or gap-filling applications, reduce reliance on autografts, and improve healing timelines.
Ultimately, the success of these technologies will depend on whether they can bridge the chasm between bench and bedside—where performance under idealized lab conditions gives way to the variability of real-world surgical practice. For now, though, their entry into human use signals that the long-promised future of bone adhesives may finally be arriving.