Enhertu nears wider EU approval as HER2 testing moves deeper into solid tumor oncology
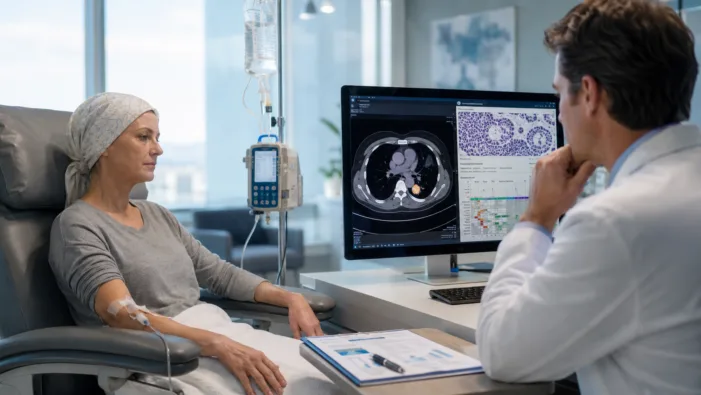
Find out how Enhertu’s EU recommendation could reshape HER2 targeted treatment for metastatic solid tumors.

Find out how Enhertu’s EU recommendation could reshape HER2 targeted treatment for metastatic solid tumors.

Datroway challenges chemotherapy in first-line TNBC. AstraZeneca and Daiichi Sankyo now face the real test: adoption, safety and access.

ENHERTU is moving earlier in breast cancer care. The bigger question is whether efficacy gains can outweigh ADC safety scrutiny.

CDH6 ADCs are heating up. OnCusp Therapeutics now needs capital discipline to turn CUSP06 promise into clinical and investor credibility.

Nuvation Bio now controls safusidenib globally. Read why the Japan deal matters for IDH1-mutant glioma, SIGMA, and the post-vorasidenib market.

Tempus AI and Daiichi Sankyo are using multimodal AI to refine ADC trial strategy and biomarker discovery. Read what this could change next.

Avacta Therapeutics (AIM: AVCT) has published preclinical data showing that its FAP-targeted payload delivery platform, FAP-Exd (AVA6103), outperforms the marketed antibody drug conjugate trastuzumab deruxtecan across three pharmacokinetic measures, with a Phase 1 clinical trial expected to begin in the first quarter of 2026. What the AI-generated comparator actually enables The methodological choice here deserves […]

SylamoreBio partners with Daiichi Sankyo to test SyLEC CNS delivery technology. Find out what this collaboration changes for brain drug development.

EMA begins review of ENHERTU plus pertuzumab for first-line HER2-positive metastatic breast cancer. Find out what this means for frontline therapy options.

AstraZeneca PLC and Daiichi Sankyo’s HER2-directed antibody-drug conjugate Enhertu (trastuzumab deruxtecan) has been granted Breakthrough Therapy Designation by the U.S. Food and Drug Administration for use in patients with HER2-positive early breast cancer who have residual invasive disease following neoadjuvant treatment and are at high risk of recurrence. The designation is based on positive results […]