Phanes Therapeutics’ spevatamig data puts cold-tumor immunotherapy back in focus at ASCO 2026
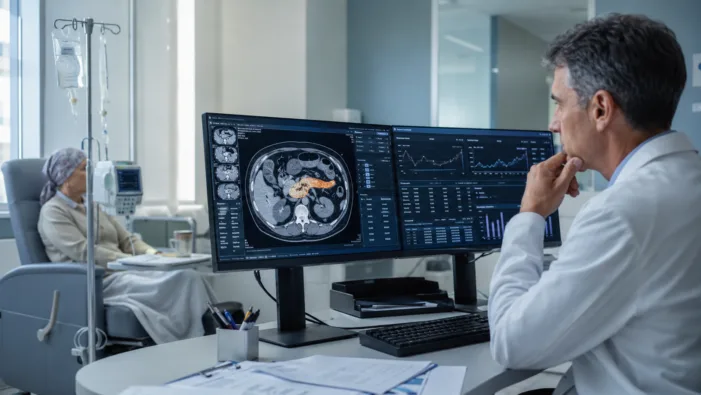
See how Phanes Therapeutics’ spevatamig data could reshape metastatic pancreatic cancer drug development after ASCO 2026.

See how Phanes Therapeutics’ spevatamig data could reshape metastatic pancreatic cancer drug development after ASCO 2026.
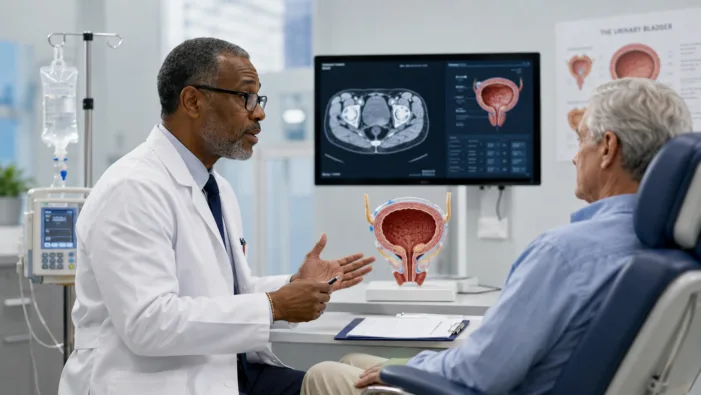
Imfinzi moves into early bladder cancer, but can AstraZeneca turn a DFS win into real-world bladder-preserving impact?

Akeso’s ivonescimab faces a defining ASCO test as lung cancer rivals watch whether PD-1/VEGF bispecifics can scale globally.

EP-NEC has no approved drug worldwide. Leads Biolabs now has to prove Opamtistomig can turn rare-tumor urgency into regulatory momentum.
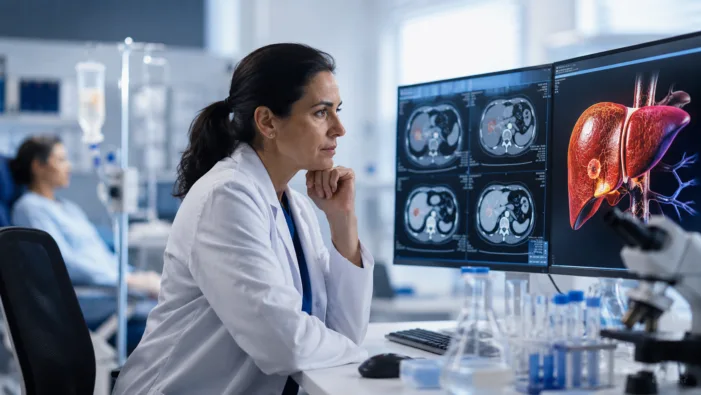
Post-ICI liver cancer has few strong options. Agenus’ BOT plus BAL data raise the next question: can durability survive larger trials?

Can-Fite BioPharma Ltd. advances Namodenoson into Phase 2b pancreatic cancer testing. Discover what this means for immunotherapy economics.
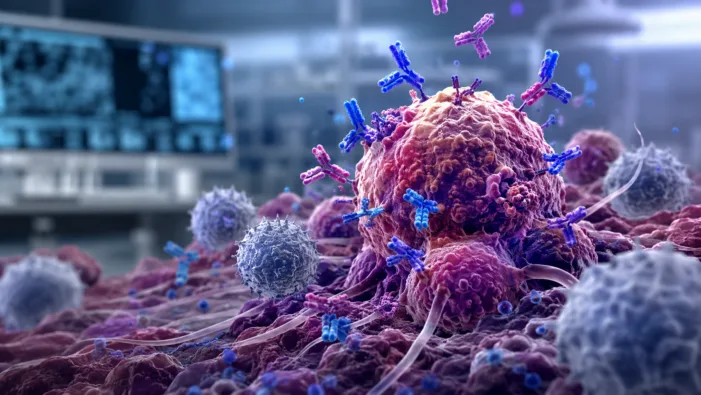
Cancer antibodies can be powerful but toxic. Ankyra’s anchored platform asks whether local retention can widen immunotherapy’s safety window.
OX40 biology has promised more than it proved. Inhibrx now needs INBRX-106 to show pembrolizumab can work harder in HNSCC.

Solid tumor T-cell engagers still face safety limits. Harbour BioMed’s HBM7004 now tests whether B7-H4 can widen that window.

Can AIM ImmunoTech Inc.’s Ampligen improve checkpoint inhibitor responses in ovarian cancer? Explore the Phase 2 findings and implications.