Can TROP2 ADCs reshape first-line NSCLC after Kelun-Biotech’s OptiTROP-Lung05 win?

A TROP2 ADC just pushed past pembrolizumab alone in first-line lung cancer. See why OptiTROP-Lung05 matters now.

A TROP2 ADC just pushed past pembrolizumab alone in first-line lung cancer. See why OptiTROP-Lung05 matters now.

Find out why Seer’s activist battle could reshape investor thinking on proteomics, diagnostics platforms and biotech value creation.

BridgeBio Pharma, Inc.’s BBP-418 wins FDA Priority Review for LGMD2I/R9. Discover what this could mean for rare muscular dystrophy treatment.

FDA approval of Hepcludex gives HDV its first U.S. therapy. Find out how this could reshape liver disease treatment and Gilead’s strategy.

Explore why Codexis, Inc.’s latest RNA manufacturing advances could influence scalability, stereochemical control, and future siRNA production economics.

Explore why the PharmAla and Jupiter Neurosciences licensing deal could reshape investor sentiment around next-generation psychedelic therapeutics.

Home care demand is rising, but margins remain tight. Enhabit’s Kinderhook exit tests whether private equity can unlock scale without care risk.

Could TN-401 change ARVC treatment strategy? Explore what Tenaya Therapeutics, Inc.’s RIDGE-1 data means for cardiac gene therapy.
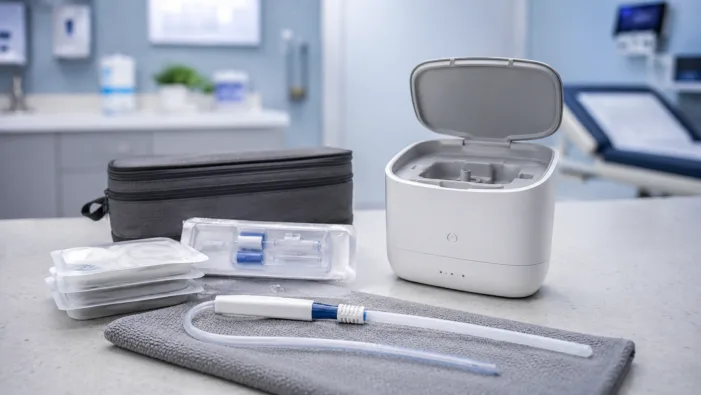
Single-use catheters finally face a regulated reuse challenge. Aurie’s FDA De Novo win could shift urology device economics.

Actuate Therapeutics, Inc. is advancing oral elraglusib after FDA IND clearance. Discover what this means for pancreatic cancer economics.