Oricell’s ASCO 2026 liver cancer data put GPC3 CAR-T back in the spotlight
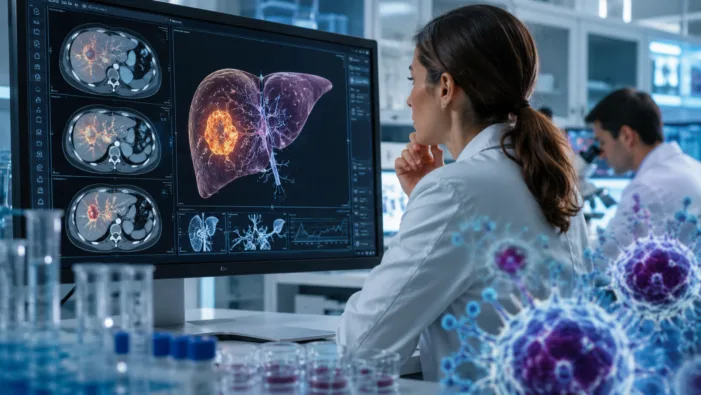
Find out how Oricell’s Ori-C101 CAR-T data could reshape expectations for late-line liver cancer treatment after ASCO 2026.

Find out how Oricell’s Ori-C101 CAR-T data could reshape expectations for late-line liver cancer treatment after ASCO 2026.
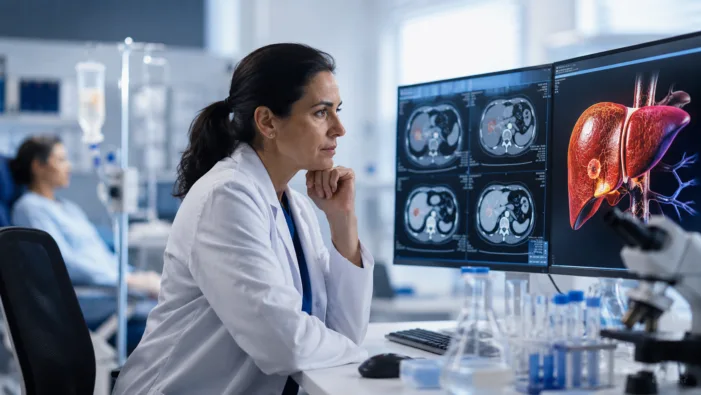
Post-ICI liver cancer has few strong options. Agenus’ BOT plus BAL data raise the next question: can durability survive larger trials?

Rznomics reported interim RZ-001 liver cancer data at AACR 2026. Read why the efficacy and safety signals could matter for hepatocellular carcinoma.

Abbisko Therapeutics won EMA orphan drug designation for irpagratinib in liver cancer. Read what this changes for FGFR4-targeted therapy.
Pilatus Biosciences earns FDA Fast Track for PLT012 in liver cancer. Read why CD36 targeting could reshape immunotherapy development.
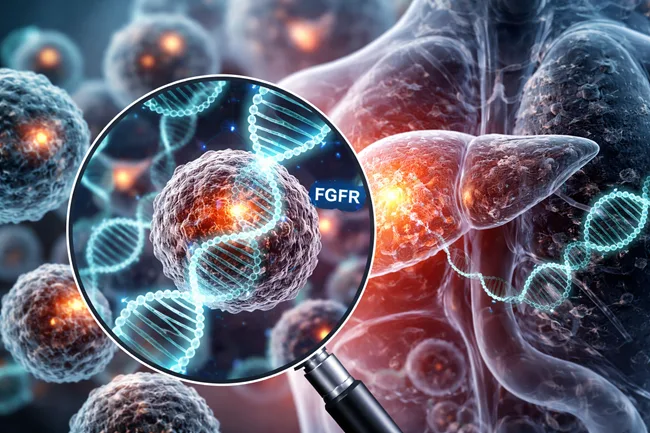
HUTCHMED (China) Limited announced on December 29, 2025, that China’s National Medical Products Administration (NMPA) has accepted its New Drug Application (NDA) for fanregratinib (HMPL-453) with priority review for second-line treatment of adult patients with intrahepatic cholangiocarcinoma harboring FGFR2 fusion or rearrangement. The NDA submission is based on a Phase II registration study in China […]

New Phase 3 subgroup data suggest Delcath’s HEPZATO KIT improves survival in patients with lower tumor burden or LDH. Learn what this means for mUM treatment.