Can Plus Therapeutics Inc. turn REYOBIQ into a differentiated platform for difficult-to-treat CNS cancers?

Can REYOBIQ change CNS cancer treatment? Discover why Plus Therapeutics Inc.’s latest leptomeningeal metastases data is drawing attention.

Can REYOBIQ change CNS cancer treatment? Discover why Plus Therapeutics Inc.’s latest leptomeningeal metastases data is drawing attention.
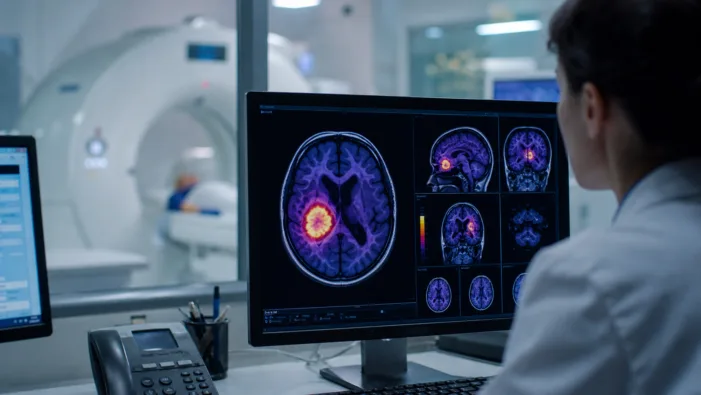
Europe needs standardized glioma PET imaging. Telix TLX101-Px now tests whether regulators and payers are ready for FET-PET access.

Telix Pharmaceuticals Limited’s TLX101-Px NDA has been accepted by the FDA. Discover what this means for glioma imaging and the September decision.

Plus Therapeutics secures orphan designation for REYOBIQ in pediatric malignant gliomas. Read what this means for trials, exclusivity, and market strategy.

Nuvation Bio now controls safusidenib globally. Read why the Japan deal matters for IDH1-mutant glioma, SIGMA, and the post-vorasidenib market.

FORE Biotherapeutics won FDA breakthrough status for plixorafenib in high-grade glioma. Read what this changes and what risks still remain.

Radiopharm Theranostics reports strong RAD 101 interim data in brain metastases imaging. Read expert analysis on clinical impact and regulatory outlook.

Curatis and Neupharma sign a CHF 83.5m Japan licensing deal for corticorelin in peritumoral brain edema. Read our analysis of what the deal means.

Find out how Adaptin Bio is preparing a Phase 1 glioblastoma trial and why its preclinical progress matters for brain cancer treatment innovation.
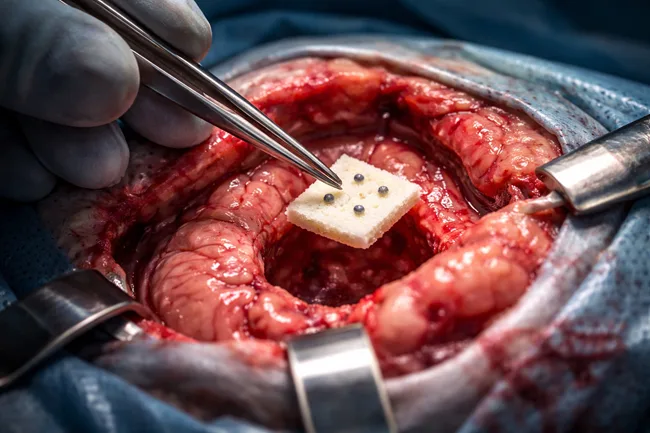
GT Medical Technologies has announced the first patient enrollments in its BRIDGES trial, a randomized clinical study assessing the use of GammaTile therapy in newly diagnosed glioblastoma. The trial introduces GammaTile at the time of initial tumor resection, marking a notable shift from the conventional glioblastoma radiation timeline, which typically delays radiation until several weeks […]