Ranok Therapeutics pushes RNK08954 forward as KRAS G12D drug development heats up

KRAS G12D was long seen as nearly untouchable. Ranok’s RNK08954 now has early clinical data, but the pivotal test is still ahead.

KRAS G12D was long seen as nearly untouchable. Ranok’s RNK08954 now has early clinical data, but the pivotal test is still ahead.
Ivonescimab showed an overall survival benefit in HARMONi-6. Read how the data could reshape first-line squamous NSCLC treatment.

Find out how SalubrisBio’s JK06 data could reshape 5T4-targeted ADC development in lung and breast cancer.
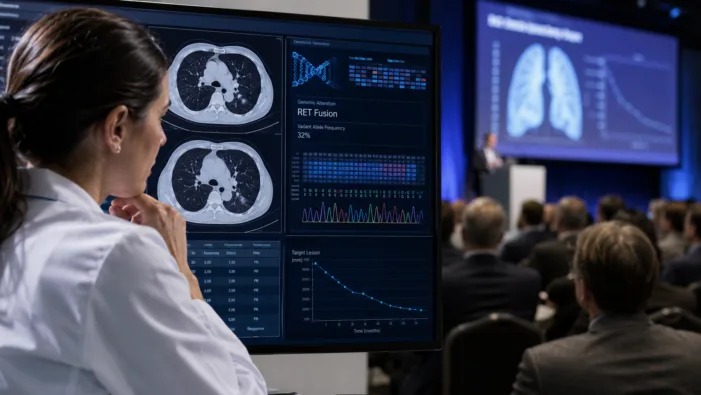
Kelun-Biotech’s RET lung cancer data looks strong, but CNS durability and global competition will decide its real impact.

Actinium’s science story is widening, but its balance sheet pressure is now impossible to ignore. ATNM-400 data may decide the next chapter.

A TROP2 ADC just pushed past pembrolizumab alone in first-line lung cancer. See why OptiTROP-Lung05 matters now.

Akeso’s ivonescimab faces a defining ASCO test as lung cancer rivals watch whether PD-1/VEGF bispecifics can scale globally.

Gilead’s Tubulis deal strengthens its ADC pipeline, but oncology execution, safety and clinical proof now decide the upside.
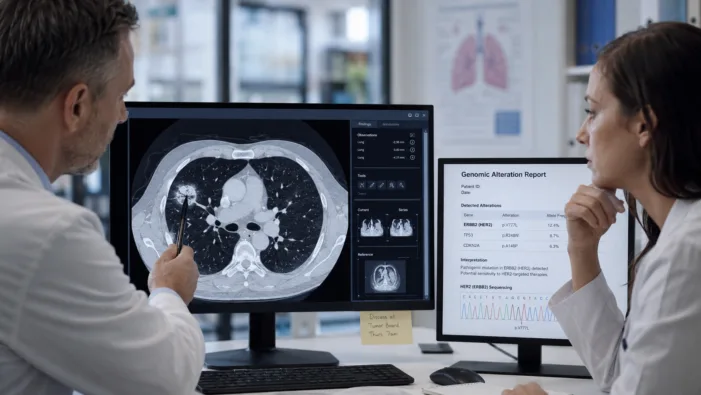
Bayer has FDA momentum in HER2-mutated lung cancer. The harder test is whether HYRNUO can move earlier without stronger survival data.

Rare cancer drugs need more than FDA approval. Nuvation Bio’s Ibtrozi test now shifts to U.S. supply reliability and launch execution.