Phoenix and Raven launch direct-to-patient generic semaglutide after Health Canada approval

Generic semaglutide is cheaper, but access is now the real battleground. Phoenix and Raven are testing Canada’s GLP-1 telehealth future.

Generic semaglutide is cheaper, but access is now the real battleground. Phoenix and Raven are testing Canada’s GLP-1 telehealth future.

Novo shelved the asset, but Cellular Intelligence sees a platform test. Parkinson’s cell therapy now faces its harder commercial trial.

Novo Nordisk has the first-mover pill. Eli Lilly has the growth aura. Oral obesity drugs are turning access into the next GLP-1 battleground.
Canada has generic Ozempic momentum. Apotex’s approval turns GLP-1 access into a pricing and supply-chain test.

Aspect Biosystems has secured a $280 million Canada-backed push in regenerative medicine. Read what it means for cellular therapy scale-up.

Novo Nordisk’s Awiqli wins FDA approval as the first once-weekly basal insulin. Read what this could change for type 2 diabetes care.

Explore how Hims & Hers and Novo Nordisk are expanding GLP-1 access and what it means for obesity care, pricing, and regulation.
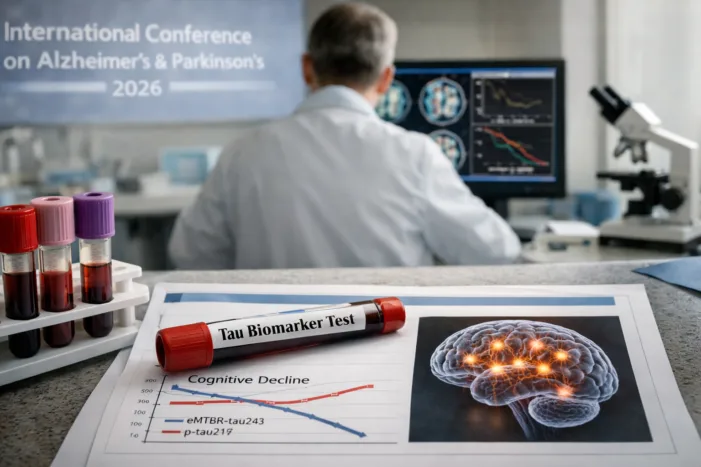
C2N’s eMTBR-tau243 plasma assay shows independent prognostic value in Phase 3 Evoke data. What this means for Alzheimer’s patient selection. Read the analysis.

Prothena earns a $50M milestone as Novo Nordisk advances coramitug in Phase 3 for ATTR cardiomyopathy. Discover what this means for amyloid therapies.

Amazon Pharmacy now delivers oral Wegovy with insurance and cash-pay options. Find out how this reshapes GLP-1 access and obesity care.