Could BeOne’s zanidatamab regimen reset expectations in HER2-positive gastroesophageal cancer?
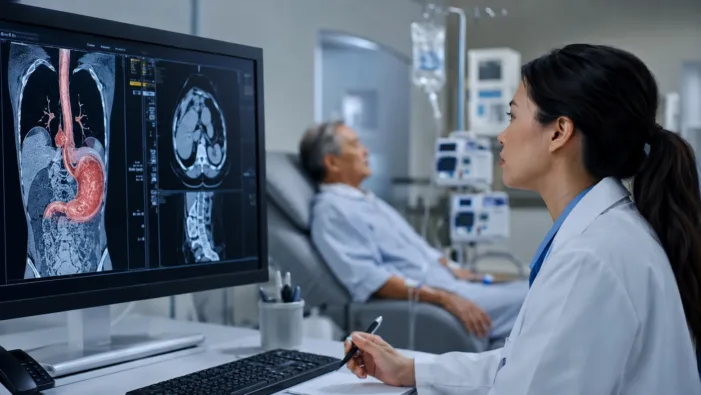
Find out how BeOne’s HERIZON-GEA data could reshape HER2-positive gastroesophageal cancer treatment after ASCO 2026.

Find out how BeOne’s HERIZON-GEA data could reshape HER2-positive gastroesophageal cancer treatment after ASCO 2026.
NUT carcinoma has few options and poor survival. Wenda Pharma’s NHWD-870 may test how fast China can move rare oncology forward.

Is Can-Fite BioPharma Ltd. carving a niche in pancreatic cancer? Explore how namodenoson’s Phase 2a data could reshape late-line treatment strategies.

Can darlifarnib overcome VEGF resistance in kidney cancer? Explore what Kura Oncology’s latest data could mean for future treatment strategies.

Read how Actuate Therapeutics’ elraglusib survival data could reshape first-line pancreatic cancer treatment and what regulators may watch next.

Setidegrasib showed early activity in KRAS G12D lung and pancreatic cancers. Read why the Phase 1 signal could matter for oncology’s next target.

IDEAYA Biosciences nears darovasertib trial data. Discover how results could reshape metastatic uveal melanoma treatment strategies.

Immutep Limited halts its TACTI-004 lung cancer trial after a futility analysis. Discover what the setback means for eftilagimod alfa and LAG-3 immunotherapy.

Citius Oncology reports early LYMPHIR plus pembrolizumab data in gynecologic cancers. Explore what the study reveals about the future of immuno-oncology strategies.
European regulators approve retifanlimab for advanced anal cancer. Discover what this means for immunotherapy treatment strategies in rare oncology today.