Can Summit and Akeso turn ivonescimab into a global lung cancer challenger?
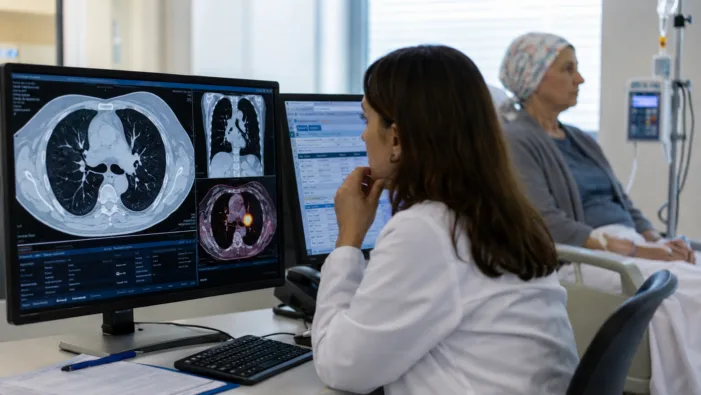
Ivonescimab showed an overall survival benefit in HARMONi-6. Read how the data could reshape first-line squamous NSCLC treatment.

Ivonescimab showed an overall survival benefit in HARMONi-6. Read how the data could reshape first-line squamous NSCLC treatment.

Read how Exelixis’ CABINET subgroup data could sharpen cabozantinib’s role in advanced neuroendocrine tumor care.

Can Massive Bio’s Reticulum Nexus suite close oncology trial access gaps? Find out how AI agents may reshape cancer trial navigation today!

CRC trials need faster evidence. KLEOS could test more therapies under one platform. Read why the model matters.

ALX Oncology has a CD47 signal. The harder test is whether ASPEN-09 can turn a tiny responder cohort into Phase 2 confidence.
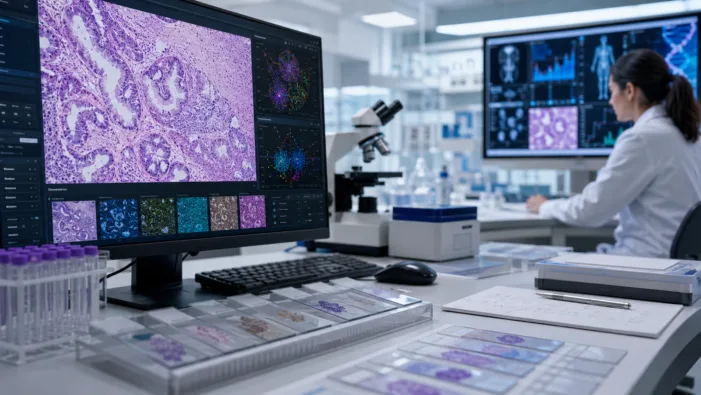
ADC trials need sharper patient selection. Daiichi Sankyo’s Waiv deal tests whether AI pathology can find response signals earlier.

FDA review is moving closer to real time. Paradigm Health’s pilot with Amgen and AstraZeneca could reset clinical trial infrastructure.

Discover how Syncromune Inc.’s SYNC-T data could reshape combination immunotherapy. Find out what this means for cancer treatment innovation.

Avenzo Therapeutics will present updated AVZO-021 Phase 1 data at ASCO 2026. Read why the CDK2 story could matter for breast cancer and beyond.

Discover how AKTX-101 could redefine TROP2 ADC resistance strategies in oncology. Read the full analysis now.