LensOne enters pivotal study as Long Bridge Medical targets a difficult gap in secondary IOL fixation
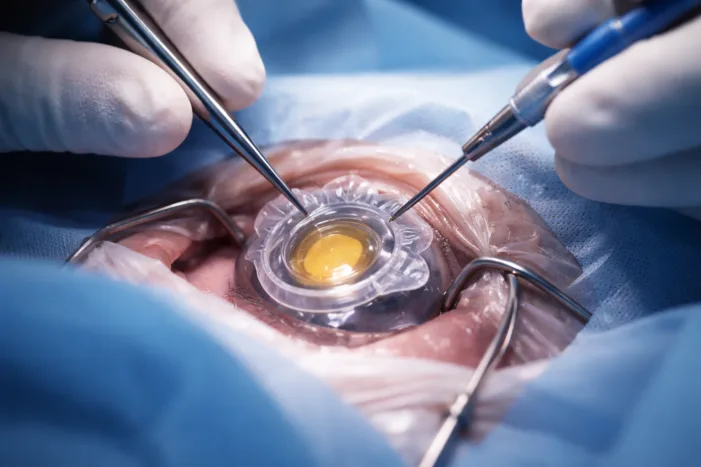
Long Bridge Medical’s LensOne has entered a pivotal FDA-cleared study. Read why this ophthalmic device trial could reshape secondary IOL fixation.

Long Bridge Medical’s LensOne has entered a pivotal FDA-cleared study. Read why this ophthalmic device trial could reshape secondary IOL fixation.

Alcon has launched TOTAL30 Multifocal for Astigmatism in the U.S. Find out why this matters for presbyopia care and contact lens adoption.