Enliven Therapeutics moves closer to Phase 3 as ELVN-001 faces its real test

ELVN-001 has early CML promise and a long cash runway. Phase 3 execution now decides whether Enliven Therapeutics can justify the hype.

ELVN-001 has early CML promise and a long cash runway. Phase 3 execution now decides whether Enliven Therapeutics can justify the hype.

Compass Pathways is set to release pivotal Phase 3 COMP360 data in treatment-resistant depression. Find out why this readout could reshape psychedelic drug development.

Discover how Idorsia Pharmaceuticals Ltd’s FDA-aligned Phase 3 plan could make lucerastat the first oral therapy for all Fabry disease patients. Read the analysis.
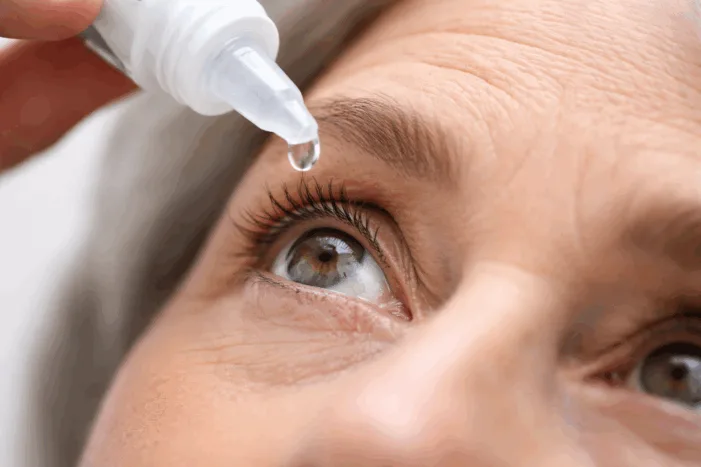
YUVEZZI becomes the first FDA-approved dual-agent eye drop for presbyopia. Find out what this means for Tenpoint Therapeutics and the future of vision care.

Find out how Cloudbreak Pharma’s Phase 2 FDA meeting reshapes late-stage risk for CBT-004 and what it signals for ophthalmic drug development.

Mirum adds brelovitug in HDV through Bluejay deal. See how this could redefine rare liver disease leadership ahead of AZURE Phase 3 results in 2026.

Johnson & Johnson (J&J) has presented new pooled data from Phase 3 clinical trials showing that CAPLYTA (lumateperone), used as an adjunctive therapy with antidepressants, significantly improved remission rates in adults with major depressive disorder compared to placebo. The results, highlighted at the 64th Annual Meeting of the American College of Neuropsychopharmacology, indicate nearly double […]

Swedish Orphan Biovitrum AB (Sobi) has entered into a definitive agreement to acquire Arthrosi Therapeutics Inc. for $950 million upfront and up to $550 million in milestone payments. The deal brings the investigational gout drug pozdeutinurad (AR882) into Sobi’s pipeline, positioning the oral URAT1 inhibitor as a potential anchor asset in a late-stage inflammatory franchise. […]

Lilly’s Jaypirca met its Phase 3 target in CLL, matching Imbruvica on response rate and showing superior safety. Could this rewire frontline BTK therapy?