Can Convergent Therapeutics define the next prostate cancer standard after Lu-PSMA therapy?
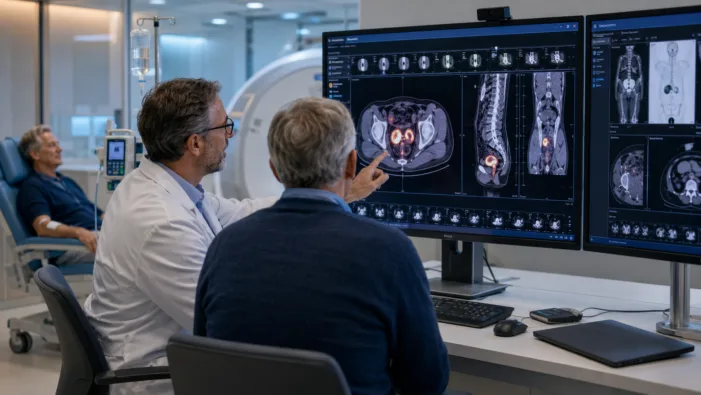
CONV01-α is targeting the post-Lu-PSMA prostate cancer gap. Convergent now faces the Phase 3 test that will decide its real value.

CONV01-α is targeting the post-Lu-PSMA prostate cancer gap. Convergent now faces the Phase 3 test that will decide its real value.

Actinium’s science story is widening, but its balance sheet pressure is now impossible to ignore. ATNM-400 data may decide the next chapter.
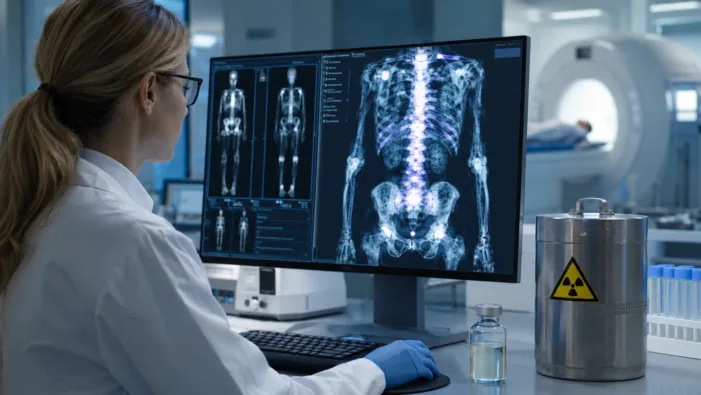
Find out how LIFT and NorthStar’s Lu-177-LT17 partnership could advance radioligand therapy for cancer bone disease today!

Find out how Niowave’s $75m Michigan facility could reshape Actinium-225 supply for targeted cancer therapies.

Radiopharma needs more than promising targets. Full-Life’s US$150m raise puts actinium-225 supply and clinical execution in focus.
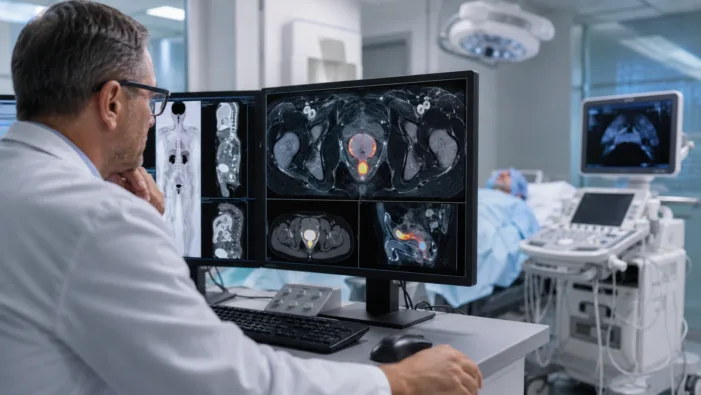
Prostate ablation is getting more precise. Telix wants PSMA-PET to become the imaging layer that helps clinicians decide where to treat.

RLT demand is rising faster than supply chains can stretch. Novartis’ Texas buildout tests whether precision oncology can scale.

Can REYOBIQ change CNS cancer treatment? Discover why Plus Therapeutics Inc.’s latest leptomeningeal metastases data is drawing attention.
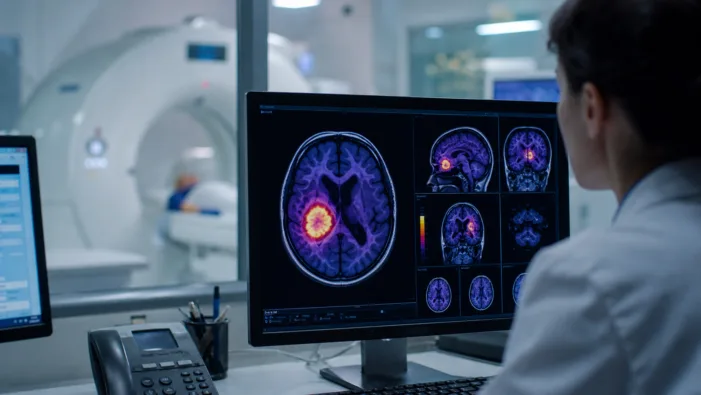
Europe needs standardized glioma PET imaging. Telix TLX101-Px now tests whether regulators and payers are ready for FET-PET access.

Find out how Juniper Biosciences is using FMISO imaging to personalize radiation therapy and reduce toxicity in head and neck cancer.