Can enicepatide and petrelintide help Genentech build a differentiated obesity franchise?

Genentech advances enicepatide and petrelintide obesity data at ADA 2026. Find out why Roche’s strategy matters now.

Genentech advances enicepatide and petrelintide obesity data at ADA 2026. Find out why Roche’s strategy matters now.
ClearNote Health will present Avantect pancreatic cancer test validation data at ASCO 2026. Find out what it could mean for early detection.

Generic semaglutide is cheaper, but access is now the real battleground. Phoenix and Raven are testing Canada’s GLP-1 telehealth future.
Canada has generic Ozempic momentum. Apotex’s approval turns GLP-1 access into a pricing and supply-chain test.

Lupin Limited has launched generic dapagliflozin in the United States. Read what it means for Farxiga, pricing pressure, and market access.

Novo Nordisk’s Awiqli wins FDA approval as the first once-weekly basal insulin. Read what this could change for type 2 diabetes care.

Explore how Hims & Hers and Novo Nordisk are expanding GLP-1 access and what it means for obesity care, pricing, and regulation.
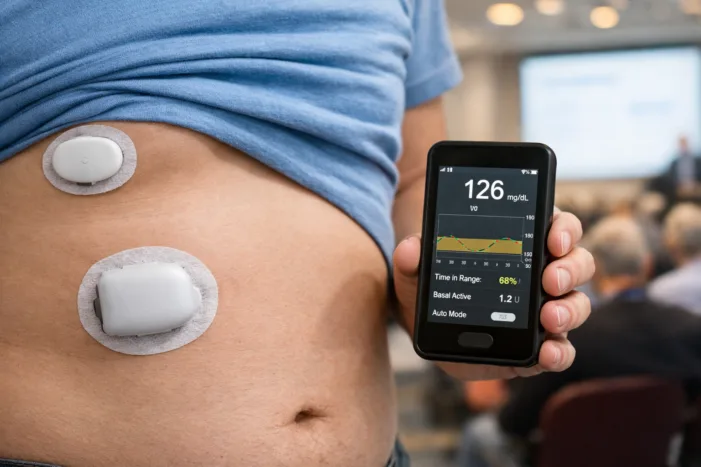
Insulet’s EVOLUTION 2 study shows 68% TIR with no boluses in type 2 diabetes. Analysis of what the FCL data means before the EVOLVE pivotal trial.

Hua Medicine’s dorzagliatin wins Hong Kong approval for Type 2 diabetes. Discover what this glucokinase activator milestone means for global diabetes treatment.

Sciwind Biosciences partners with Pfizer China on ecnoglutide. Explore what this GLP-1 deal changes for obesity, diabetes, and China’s drug market.