Is Butterfly Medical’s prostate retraction device the next challenger in minimally invasive BPH treatment?
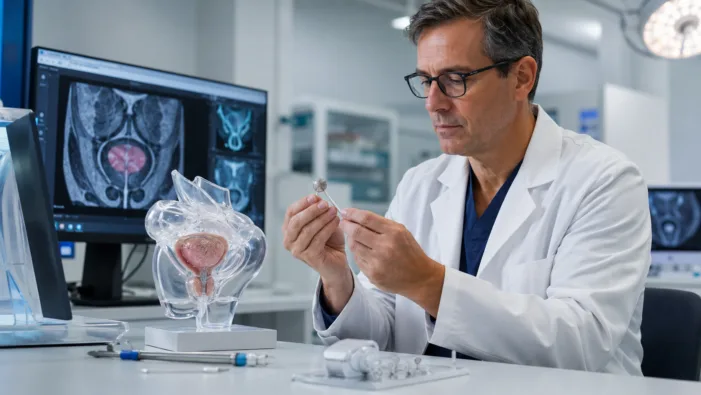
BPH care needs options beyond pills and surgery. Butterfly Medical’s $21m raise puts its reversible prostate implant in the spotlight.

BPH care needs options beyond pills and surgery. Butterfly Medical’s $21m raise puts its reversible prostate implant in the spotlight.

Neuspera Medical’s 12-month pivotal SNM trial shows 94% responder rates and zero infections. Analysing what this means for the SNM market. Read more.

Francis Medical launches commercial Vanquish procedures for prostate tissue ablation. Explore what this means for minimally invasive prostate cancer treatment.
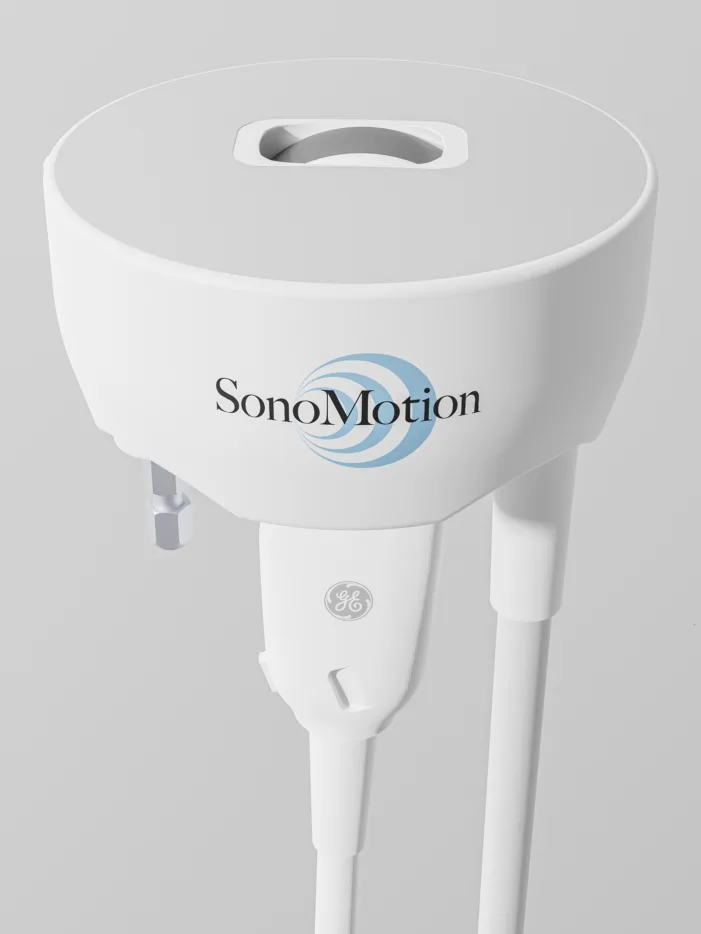
SonoMotion’s Break Wave lithotripsy device wins FDA clearance for non-invasive kidney stone treatment. Find out what this changes for urology practices.