Is Avvio Medical building the next outpatient frontier in kidney stone and ureteral stone therapy?

Ureteral stones need faster care. Avvio Medical’s outpatient device tests whether microbubble lithotripsy can ease OR bottlenecks.

Ureteral stones need faster care. Avvio Medical’s outpatient device tests whether microbubble lithotripsy can ease OR bottlenecks.
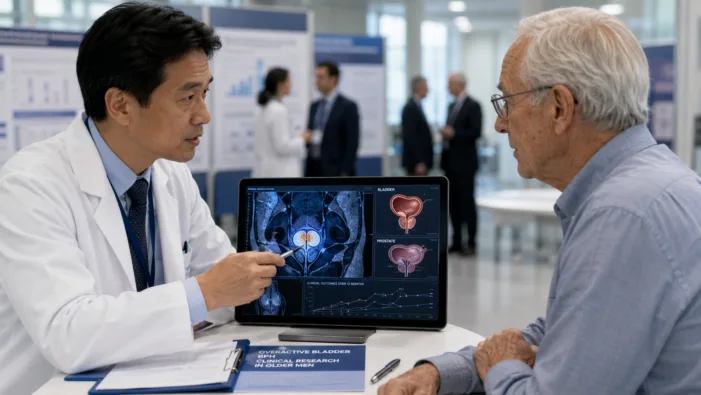
Older men with OAB and BPH remain hard to treat. Sumitomo Pharma America’s vibegron data may sharpen the next urology market test.

Bayer has stronger NUBEQA mCSPC data. The tougher test is whether clinicians view ARASEC as confirmatory or merely supportive.

UUI devices have clinical momentum. BlueWind Medical’s next challenge is proving Revi can scale through access, reimbursement, and workflow adoption.
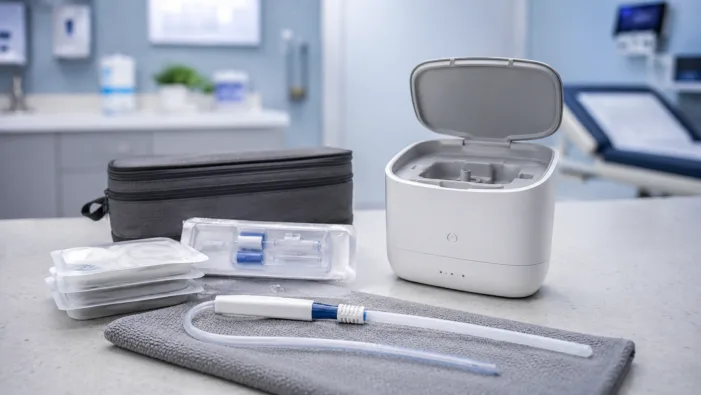
Single-use catheters finally face a regulated reuse challenge. Aurie’s FDA De Novo win could shift urology device economics.

PanGIA Biotech reported 97.8% sensitivity in a urine-based prostate cancer study. Read what the data changes and what risks still remain.

ImmunityBio has resubmitted its supplemental biologics license application to the U.S. Food and Drug Administration for ANKTIVA (nogapendekin alfa inbakicept) in combination with Bacillus Calmette-Guerin for BCG-unresponsive non-muscle invasive bladder cancer with papillary tumors, after the agency requested updated long-term efficacy data in March 2026. The resubmission follows a regulatory exchange that began in January […]