Is PMS relief becoming a consumer wellness category? Bayer’s Midol in Motion campaign suggests so

PMS relief is moving beyond pain pills. Bayer’s Midol campaign ties symptom care to movement, creators, and menstrual wellness.

PMS relief is moving beyond pain pills. Bayer’s Midol campaign ties symptom care to movement, creators, and menstrual wellness.
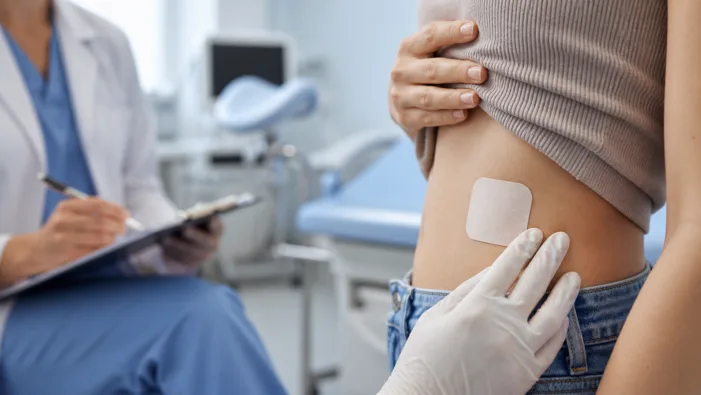
Viatris has patch expertise. FDA scrutiny now decides whether lower estrogen and adhesion data can turn MR-100A-01 into a real growth lever.

Is Noema Pharma AG building a menopause therapy platform? Discover how cendifensine could reshape non-hormonal treatment strategies

Sun Pharma gets global scale with Organon. The harder test is whether women’s health, biosimilars, and debt discipline can move together.

Vistagen advances refisolone for menopausal hot flashes after FDA clearance. Find out what this means for non-hormonal treatment options.

Uqora has entered Target stores and Target.com. Find out what this expansion means for urinary health retail and women’s wellness growth.

AbCellera will reveal Phase 1 ABCL635 data on May 11. Read why this menopause drug update could matter for investors and women’s health watchers.

Pendulum Therapeutics has expanded its Mayo Clinic collaboration into women’s health and dermatology. Read what this could mean for microbiome medicine.

Peri has launched a perimenopause wearable, but can symptom tracking become clinical-grade insight? Read the deeper industry analysis now.
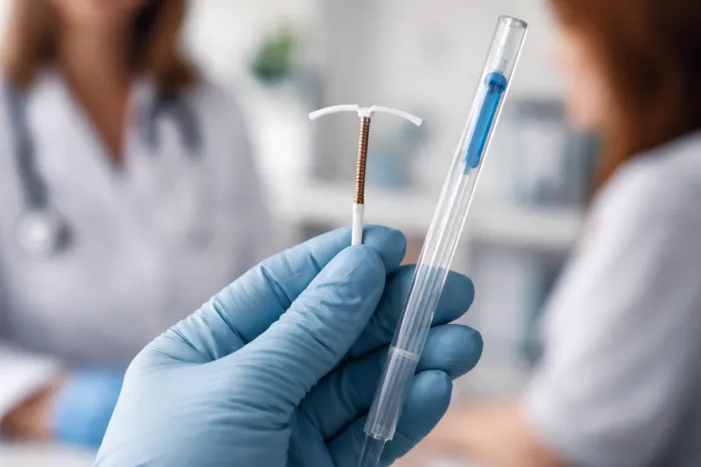
Organon & Co. licenses MIUDELLA copper IUD. Explore what this means for hormone-free contraception and LARC competition. Read the analysis.