Abbott has received U.S. Food and Drug Administration clearance and CE Mark approval for Ultreon 3.0 Software, its next-generation artificial intelligence-powered coronary imaging platform for optical coherence tomography-guided percutaneous coronary intervention. The regulatory milestone gives Abbott a stronger position in complex coronary artery disease treatment, where physicians are under pressure to improve stent planning, reduce procedural uncertainty, and make intravascular imaging easier to use in routine and high-risk cases.
Why Abbott’s Ultreon 3.0 clearance matters for the future of AI-guided coronary intervention
The significance of Ultreon 3.0 is not simply that Abbott has added artificial intelligence to an existing imaging workflow. The more important development is that AI-based plaque assessment, vessel measurement, stent sizing support, and post-procedure review are being pushed into a single interventional platform rather than remaining as separate interpretive steps. For physicians treating complex coronary disease, that integration could matter because procedural decisions often need to be made quickly while the patient is already on the table.
Optical coherence tomography has long offered high-resolution intravascular imaging, but adoption has faced practical friction. It can generate rich information, yet that information must be converted into clinical decisions during a time-sensitive procedure. Abbott’s updated platform attempts to close that gap by using automated insights to support lesion assessment, stent placement, and post-stent evaluation. The strategic question is whether automation can make advanced imaging feel less like a specialist add-on and more like a natural part of complex PCI workflow.
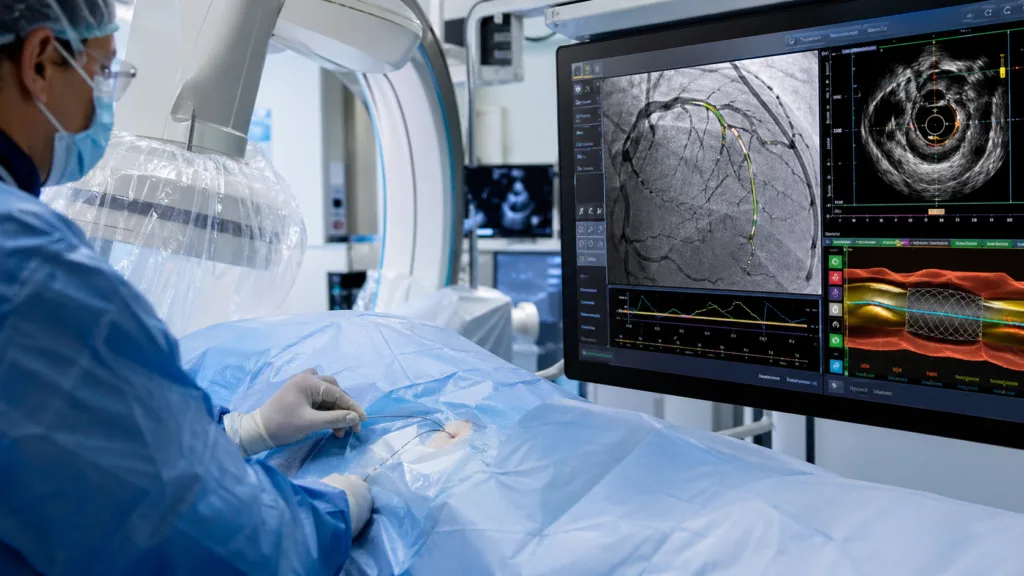
That is where the competitive importance of Ultreon 3.0 becomes clearer. Medical device manufacturers are no longer competing only on catheter hardware, imaging clarity, or stent portfolios. They are increasingly competing on procedural intelligence. The manufacturer that can help cardiologists interpret images faster, standardize decision-making, and reduce avoidable variation may gain influence not only in cath labs but also in hospital purchasing conversations. For Abbott, Ultreon 3.0 strengthens the link between its vascular device portfolio and its broader push into AI-enabled clinical support.
How Ultreon 3.0 could change stent planning in complex coronary artery disease
Complex PCI is particularly suited to AI-assisted imaging because the procedure often involves difficult lesions, calcified plaque, long blockages, bifurcations, or patients who may need multiple stents. In such cases, the difference between approximate stent placement and image-guided precision can be clinically meaningful. Ultreon 3.0 is designed to help physicians understand plaque type, lesion geometry, vessel size, and stent positioning before and after intervention.
The most relevant clinical promise is consistency. Experienced interventional cardiologists may already be skilled at interpreting OCT images, but wider use depends on making interpretation faster, easier, and less dependent on individual operator experience. AI-powered guidance may help reduce variability by converting detailed image data into structured procedural insights. That does not replace physician judgment, but it can change the threshold for using advanced imaging in cases where the operator might otherwise rely more heavily on angiography.
The risk is that better imaging does not automatically translate into better outcomes unless it changes decisions in a measurable way. Hospitals and clinicians will want to see whether AI-guided OCT use improves stent expansion, reduces malapposition, lowers repeat revascularization, or meaningfully affects adverse cardiovascular events. The platform’s promise is strong from a workflow and procedural planning perspective, but the broader adoption story will depend on how convincingly Abbott can connect those capabilities to real-world clinical and economic value.
Why OCT’s clinical value still depends on workflow, contrast use, and physician adoption
Ultreon 3.0’s one-second OCT pullback is an important workflow element because speed matters in interventional cardiology. Rapid image acquisition can reduce procedural interruption and make advanced imaging easier to incorporate into complex cases. The platform’s ability to support low or zero contrast imaging is also relevant because contrast exposure can be a concern in patients with kidney disease, a population that overlaps meaningfully with coronary artery disease.
The contrast issue may become one of the more practical adoption drivers. Intravascular imaging is often discussed in terms of resolution and precision, but cath labs must also manage patient comorbidities, procedure time, contrast burden, reimbursement realities, and staff training. A system that reduces contrast dependency while improving procedural planning may be attractive in medically complex patients, especially when physicians are trying to balance diagnostic clarity with renal risk.
However, adoption still depends on whether the technology simplifies the procedure enough to justify its use. If AI automation adds confidence but also adds setup time, cost, or training complexity, uptake may remain concentrated in high-volume centers and specialist operators. Abbott’s challenge will be to make Ultreon 3.0 feel operationally efficient, not merely technologically impressive. In medtech, the best clinical tool can still stall if it does not fit smoothly into the rhythm of real-world hospital care.
What is genuinely new versus incremental in Abbott’s Ultreon 3.0 platform
Ultreon 3.0 appears to be an incremental platform evolution with potentially meaningful adoption implications. The underlying ingredients, OCT imaging, PCI guidance, plaque visualization, and AI-assisted interpretation, are not entirely new concepts. What changes is the level of integration and the attempt to make AI-automated insights more directly actionable during the procedure.
That distinction matters because medtech innovation is often less about inventing a completely new category and more about removing barriers that prevented a known technology from scaling. OCT has a strong clinical rationale, but broader use has depended on making the data easier to interpret and more connected to treatment planning. By positioning Ultreon 3.0 as a fast, integrated, AI-enabled system, Abbott is trying to move OCT from a high-resolution imaging modality toward a more complete decision-support platform.
The unresolved question is whether clinicians will view the platform as a genuine step-change or as a useful upgrade within an established imaging category. That perception will shape adoption. If physicians see Ultreon 3.0 as reducing cognitive load and improving procedural confidence, the clearance could become commercially meaningful. If they see it mainly as a software enhancement, the impact may be more gradual and tied to replacement cycles, training programs, and institutional preference for image-guided PCI.
How Abbott’s AI imaging push fits into the wider cardiovascular device market
Abbott’s move reflects a broader shift in cardiovascular devices, where hardware innovation is increasingly being paired with software, imaging, and artificial intelligence. The future of interventional cardiology may be less about standalone devices and more about ecosystems that guide treatment from diagnosis through intervention and follow-up assessment. Ultreon 3.0 fits that direction because it attempts to turn intravascular imaging into a procedural intelligence layer.
For Abbott, the platform also reinforces a strategic advantage that extends beyond a single software clearance. A strong coronary imaging tool can support the broader vascular franchise by influencing how physicians plan interventions and evaluate results. If imaging helps determine stent size, placement, and post-procedure adequacy, then the company offering the imaging workflow can become more embedded in clinical decision-making.
The competitive pressure will not be limited to one rival or one technology class. Intravascular ultrasound, OCT, angiography-based physiology tools, computational planning software, and AI-enabled cath lab platforms are all converging around the same objective: better procedural decisions with less uncertainty. Abbott’s task is to show that Ultreon 3.0 offers enough clarity, speed, and usability to justify its place in that crowded decision-support stack.
What clinicians and hospitals are likely to watch after Ultreon 3.0’s regulatory milestone
Clinicians are likely to focus on whether Ultreon 3.0 improves confidence in lesion preparation, stent sizing, and post-stent optimization in complex cases. These are practical questions, not abstract technology questions. Interventional cardiologists will want to know whether the platform helps them make better decisions faster, especially in difficult anatomy or high-risk patients.
Hospitals will look at a different but related set of questions. They will consider whether the platform can improve procedural efficiency, support standardization, reduce variability across operators, and strengthen outcomes documentation. Reimbursement and cost justification will matter, especially in systems where advanced imaging must compete with other cath lab priorities. The more Abbott can connect Ultreon 3.0 to measurable procedural value, the stronger the commercial argument becomes.
Regulators and industry observers will also watch how AI-enabled procedural tools evolve after clearance. In cardiovascular intervention, AI is moving from retrospective image review toward real-time decision support. That creates opportunities, but it also raises questions about validation, user trust, training, liability, and the boundary between assistance and clinical judgment. Ultreon 3.0’s progress may therefore become part of a larger debate about how much procedural decision-making can be safely and effectively automated.
Why Abbott’s biggest opportunity may be standardizing precision PCI rather than replacing clinician judgment
The most realistic opportunity for Ultreon 3.0 is not that AI replaces the interventional cardiologist. The stronger proposition is that AI could make precision PCI more consistent, more accessible, and easier to perform at scale. In that sense, the platform’s value lies in augmenting physician judgment rather than attempting to override it.
This is an important distinction for adoption. Clinicians tend to resist tools that appear to reduce their control but are more open to tools that reduce friction, improve confidence, and surface relevant information at the right moment. Ultreon 3.0 appears designed around that second model. It supports the physician by helping convert OCT image detail into actionable procedural guidance.
The limitation is that AI-assisted consistency must be proven in everyday practice, not only in controlled demonstrations or expert-center use. Complex PCI is heterogeneous, patient anatomy varies widely, and procedural decisions often involve multiple trade-offs. Abbott has cleared an important regulatory and commercial hurdle, but the deeper test will come from real-world use: whether Ultreon 3.0 can help physicians treat complex coronary disease with greater precision without adding complexity to an already demanding procedure.
What Abbott’s Ultreon 3.0 signals about the next phase of AI in medical devices
Ultreon 3.0 signals that AI in medical devices is entering a more practical phase. The early story around artificial intelligence in healthcare often centered on diagnosis, prediction, and administrative automation. In interventional cardiology, the opportunity is more immediate: assist a physician during a procedure where every measurement, placement decision, and post-procedure assessment can affect the final result.
That makes Abbott’s clearance strategically important even if adoption unfolds gradually. AI-guided OCT is not just another software feature. It is part of a larger movement toward image-guided, data-assisted, precision intervention. If that model succeeds, future cath labs may increasingly rely on integrated platforms that combine imaging, automated interpretation, device selection support, and outcome assessment.
For Abbott, Ultreon 3.0 expands its role in cardiovascular care from device supplier to procedural intelligence partner. For clinicians, the platform could make advanced coronary imaging faster and more actionable. For hospitals, the question will be whether those benefits translate into consistent procedural value. The technology has cleared the regulatory gate in the United States and Europe. The harder test now begins inside cath labs, where adoption will depend on trust, usability, evidence, and whether AI can quietly make a difficult procedure feel more precise.