Ribo’s coronary artery disease filing highlights the next frontier for RNA-based cardiovascular drugs

Ribo’s RBD1119 Phase 2 filing could test whether siRNA drugs can reduce coronary artery disease risk without bleeding trade-offs.

Ribo’s RBD1119 Phase 2 filing could test whether siRNA drugs can reduce coronary artery disease risk without bleeding trade-offs.

Can Eli Lilly and Company’s VERVE-102 transform cholesterol treatment through one-time gene editing? Explore the risks, data, and implications.
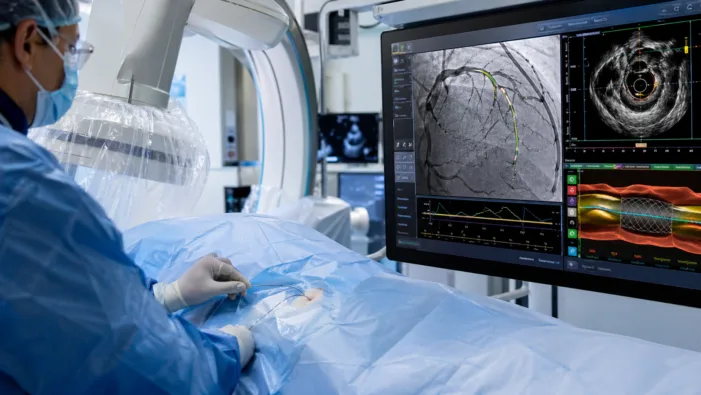
AI can now guide coronary imaging in real time. Abbott’s Ultreon 3.0 puts precision PCI closer to routine clinical workflow.

Find out how Reflow Medical’s Cora Flex and Cora Force launch could reshape complex coronary intervention workflows.

CorVista Health presents machine-learned non-invasive INOCA detection data at ACC.26. Analysis of what this means for cardiology and women’s heart care. Read more.

Cleerly’s INVICTUS registry shows AI-QCT matches IVUS in coronary plaque quantification. Read the clinical analysis.