Can Exelixis defend its oncology franchise with deeper CABINET evidence?

Read how Exelixis’ CABINET subgroup data could sharpen cabozantinib’s role in advanced neuroendocrine tumor care.

Read how Exelixis’ CABINET subgroup data could sharpen cabozantinib’s role in advanced neuroendocrine tumor care.

Can Massive Bio’s Reticulum Nexus suite close oncology trial access gaps? Find out how AI agents may reshape cancer trial navigation today!

Mirum Pharmaceuticals’ EASL 2026 data deepen its rare liver disease strategy. Find out what changes for PSC, HDV and PFIC care.

PeproMene Bio’s PMB-CT01 data raise fresh questions for post-CD19 CAR T lymphoma care. Find out what the early signal means.

CRC trials need faster evidence. KLEOS could test more therapies under one platform. Read why the model matters.

GLP-1 drugs dominate obesity care, but Kalohexis is testing a different pathway. 710GO could shift the next weight loss race.
Atypical EGFR lung cancer lacks clear first-line answers. Rybrevant plus Lazcluze may change that, but trial size still matters.

Menarini’s elacestrant strategy is shifting from monotherapy to combinations. Can ELEVATE data support a broader breast cancer role?
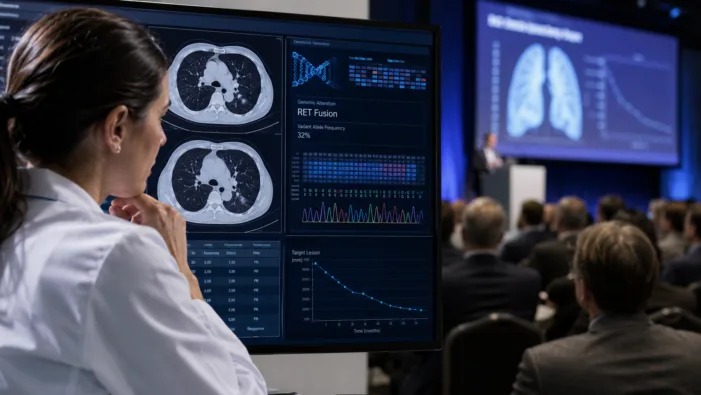
Kelun-Biotech’s RET lung cancer data looks strong, but CNS durability and global competition will decide its real impact.
HBOT demand is rising beyond hospitals. MD Hyperbaric’s clinic expansion tests whether recovery care can scale without losing clinical rigor.