KARL STORZ has received clearance from the United States Food and Drug Administration for the FIVE S 6.5 sterile single-use bronchoscope, a device intended for bronchoscopy, airway inspection, and lavage procedures in critical care settings. The clearance positions the bronchoscope for use by intensivists, respiratory therapists, and clinicians managing high-risk patients requiring rapid airway visualization and secretion clearance in intensive care environments.
The regulatory milestone adds another product to the airway management portfolio of the Germany-based endoscopy manufacturer at a time when hospitals are increasingly evaluating disposable bronchoscopy platforms. While the approval itself is incremental in regulatory terms, it reflects deeper structural shifts in how hospitals are approaching infection control, device availability, and ICU workflow efficiency.
How the FDA clearance highlights the accelerating shift toward disposable bronchoscopy in hospital airway management
Bronchoscopy has traditionally relied on reusable scopes that must undergo rigorous reprocessing between procedures. These cleaning protocols involve specialized sterilization equipment, trained personnel, and strict compliance procedures to prevent contamination. Although such systems are widely used, they can create operational bottlenecks in environments where immediate device access is required.
Single-use bronchoscopes are emerging as a solution to these logistical challenges. Disposable devices eliminate the need for reprocessing and allow hospitals to maintain ready-to-use equipment in emergency departments, operating rooms, and intensive care units. For clinicians facing respiratory emergencies, immediate access to a sterile bronchoscope can reduce delays during airway management procedures.
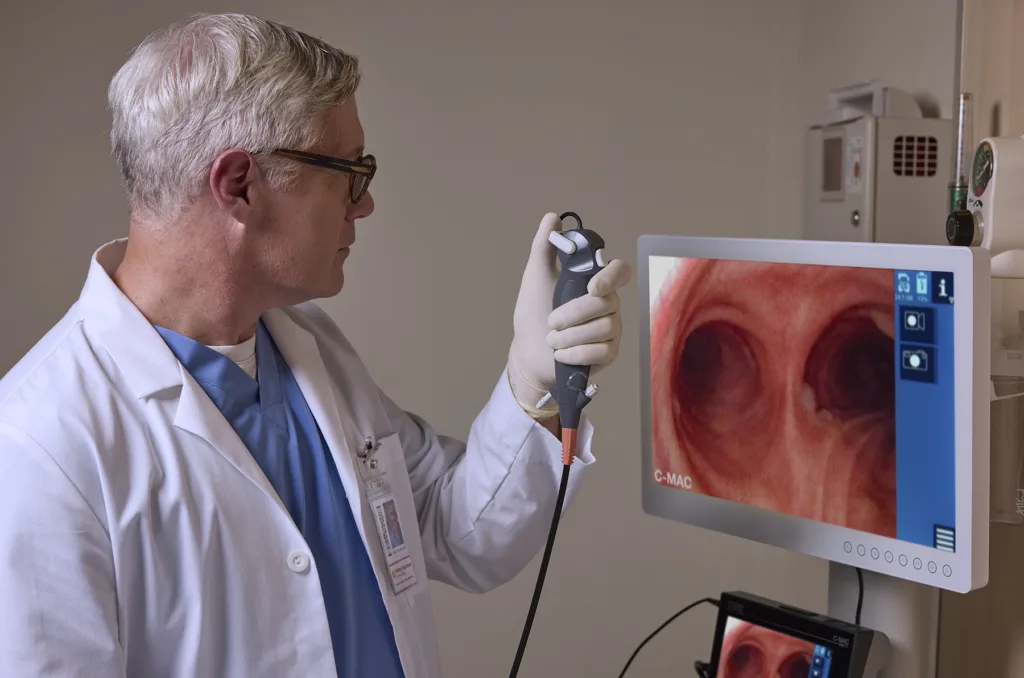
Industry observers note that the transition toward disposable bronchoscopy accelerated during the COVID-19 pandemic, when infection control concerns heightened scrutiny of reusable endoscopic equipment. The pandemic highlighted the operational advantages of devices that could be deployed rapidly without the risk of cross-contamination between patients.
The clearance of the FIVE S 6.5 bronchoscope therefore aligns with a broader market movement rather than introducing an entirely new clinical category. The medical device manufacturer appears to be positioning the device as part of a growing ecosystem of single-use airway technologies designed to support critical care workflows.
Why suction capability and working channel design remain crucial in ICU bronchoscopy procedures
One of the most technically significant elements of the FIVE S 6.5 bronchoscope is its 3.0 millimeter working channel, which enables strong suction capability while allowing the use of bronchoscopic instruments. In critical care bronchoscopy, suction performance is often a defining functional parameter.
Patients in intensive care frequently present with thick airway secretions, blood, or other obstructions that can compromise ventilation. Clearing these secretions quickly is essential to restoring airway patency and enabling adequate oxygenation. Devices with insufficient suction performance may limit the effectiveness of bronchoscopic interventions in these scenarios.
Bench testing conducted by the manufacturer indicated that the bronchoscope could clear simulated mucus volumes rapidly through its working channel. While laboratory performance metrics do not necessarily translate directly to clinical outcomes, suction efficiency remains a key factor clinicians consider when selecting bronchoscopy equipment.
The engineering challenge for single-use devices is maintaining performance while controlling manufacturing costs. Disposable bronchoscopes must achieve sufficient suction, articulation, and visualization capabilities without the durability requirements associated with reusable scopes. This balance between performance and cost efficiency often determines how competitive a device will be in hospital procurement decisions.
How integration with existing visualization systems may influence hospital adoption decisions
Another strategic aspect of the FIVE S 6.5 bronchoscope is its compatibility with the C-MAC visualization system widely used for video laryngoscopy and airway procedures. Integration with an existing platform can significantly reduce barriers to adoption in hospital settings.
Healthcare facilities typically avoid introducing new medical technologies that require extensive infrastructure changes or staff retraining. Devices that connect to equipment already in use can therefore move through procurement processes more easily.
In airway management, visualization platforms often serve multiple clinical functions, including laryngoscopy, bronchoscopy, and intubation support. A bronchoscope that operates on the same monitor used for these procedures can streamline clinical workflows while minimizing equipment redundancy.
Industry observers note that platform compatibility can create strategic advantages for device manufacturers. Hospitals that have already standardized around a specific visualization platform may be more inclined to adopt compatible devices within the same ecosystem.
In this context, the integration of the FIVE S 6.5 bronchoscope with the C-MAC system may be as strategically important as the device’s technical specifications. Platform alignment can encourage hospitals to view the bronchoscope as an extension of an existing airway management infrastructure rather than a separate product category.
What the clearance suggests about competition in the single-use bronchoscopy market
The disposable bronchoscopy segment has become one of the fastest growing areas within airway management technology. Several companies have developed single-use bronchoscopes designed specifically for intensive care and anesthesia environments.
Companies such as Ambu have established significant market presence with disposable bronchoscopy platforms that emphasize convenience and infection control. Other medical device manufacturers have entered the market with their own single-use designs aimed at addressing ICU workflow challenges.
For KARL STORZ, the clearance of the FIVE S 6.5 bronchoscope represents a strategic expansion into this competitive segment. Historically known for reusable endoscopy systems used in surgical procedures, the manufacturer appears to be broadening its product portfolio to address the growing demand for disposable devices.
Entering the single-use bronchoscopy market may allow the manufacturer to compete more directly in critical care settings where disposable scopes are gaining traction. However, differentiation will likely depend on several factors including image quality, suction performance, ergonomic design, and compatibility with hospital infrastructure.
Clinicians tracking the field believe device selection will increasingly depend on practical performance factors rather than incremental feature differences. Hospitals are likely to evaluate how effectively disposable bronchoscopes integrate into ICU workflows and whether they offer meaningful advantages over existing options.
Why infection control and workflow efficiency are reshaping bronchoscopy technology development
Infection prevention remains one of the strongest drivers of innovation in bronchoscopy equipment. Reusable endoscopes require meticulous reprocessing procedures to eliminate the risk of pathogen transmission between patients. Even with strict protocols, occasional contamination events have prompted regulatory scrutiny of endoscope cleaning practices.
Disposable bronchoscopes eliminate the need for reprocessing and reduce the potential for cross-contamination. Each device arrives sterile and is discarded after use, simplifying infection control protocols and reducing reliance on complex sterilization infrastructure.
For hospitals managing large numbers of critically ill patients, the operational advantages can be significant. Disposable devices reduce turnaround times between procedures and ensure that a sterile bronchoscope is immediately available when required.
At the same time, the transition toward single-use devices raises new considerations for healthcare systems. Hospitals must weigh infection prevention benefits against the cost of disposable equipment and the environmental impact associated with increased medical waste.
Regulatory watchers suggest that future device development may increasingly focus on balancing these competing priorities. Innovations in recyclable materials or more efficient manufacturing processes could become important factors in the next generation of disposable endoscopy products.
What clinicians and regulators may monitor as single-use bronchoscopy adoption expands
As the adoption of disposable bronchoscopes continues to expand, clinicians and healthcare administrators will likely evaluate several key factors. One central question is whether single-use devices can consistently match the performance of reusable bronchoscopes in complex clinical procedures.
Reusable scopes have historically offered superior optical systems, greater instrument compatibility, and enhanced maneuverability. Although disposable devices have improved significantly in recent years, their performance limitations remain a subject of ongoing evaluation.
Economic considerations will also play a role in shaping adoption patterns. Disposable bronchoscopes may reduce sterilization costs and operational complexity, but the per-procedure expense can be higher than reusable alternatives in certain clinical settings.
Environmental sustainability is another emerging issue. Healthcare systems are increasingly under pressure to reduce waste generated by single-use medical products. As disposable bronchoscopy expands, manufacturers may face growing expectations to address environmental concerns associated with device disposal.
Why airway management technology is becoming a strategic focus for medical device manufacturers
Airway management devices occupy a critical position within acute care medicine. Clinicians rely on bronchoscopes, laryngoscopes, and related tools to secure airways, diagnose pulmonary conditions, and perform life-saving interventions.
As patient populations become older and more medically complex, demand for reliable airway technologies is expected to grow. Hospitals are increasingly seeking devices that combine visualization, suction capability, and procedural flexibility within integrated platforms.
For medical device manufacturers, this evolving clinical landscape creates opportunities to develop comprehensive airway management ecosystems rather than standalone products. Visualization monitors, disposable scopes, and accessory instruments can be designed to function together within unified procedural workflows.
The FDA clearance of the FIVE S 6.5 bronchoscope therefore represents more than a single device launch. It reflects a broader industry effort to align airway management technologies with the operational realities of modern critical care environments.