Cairn Surgical, Inc. has reported pivotal trial results for its investigational Breast Cancer Locator System, a patient-specific surgical guidance device designed to improve tumor localization during breast-conserving surgery. The international randomized trial met its primary endpoint by showing a lower positive margin rate than conventional wire localization, positioning the Breast Cancer Locator as a potential alternative to one of the most established but imperfect workflows in non-palpable breast cancer surgery.
Why Cairn Surgical’s trial matters for a procedure still shaped by positive margin risk
The most important signal from the pivotal trial is not simply that Cairn Surgical reported statistical success against conventional wire localization. It is that the Breast Cancer Locator is targeting a persistent operational problem in breast-conserving surgery, where surgeons must remove enough tissue to clear the tumor while preserving as much healthy breast tissue as possible. Positive margins remain one of the most consequential failure points in lumpectomy because they can trigger additional surgery, delay adjuvant treatment, increase patient anxiety, and add cost to the care pathway.
Cairn Surgical said the pivotal study showed a 32% reduction in positive margin rate across all patients and a 34% reduction in re-excisions when compared with conventional wire localization. That is clinically meaningful because re-excision is not a minor inconvenience in breast cancer care. A second surgery can alter cosmetic outcomes, strain surgical capacity, increase payer burden, and complicate patient experience at a moment when healthcare systems are already trying to standardize more efficient cancer pathways.
However, the unresolved question is how strongly the trial result will translate into routine practice across different hospital settings. A reduction in positive margins is valuable, but adoption will depend on whether surgeons can integrate the device without adding excessive preoperative complexity, imaging dependency, planning time, manufacturing logistics, or scheduling friction. In surgical oncology, a better guidance tool only scales if it fits the rhythm of operating rooms, radiology departments, pathology workflows, and reimbursement systems.
How the Breast Cancer Locator challenges the limits of conventional wire localization
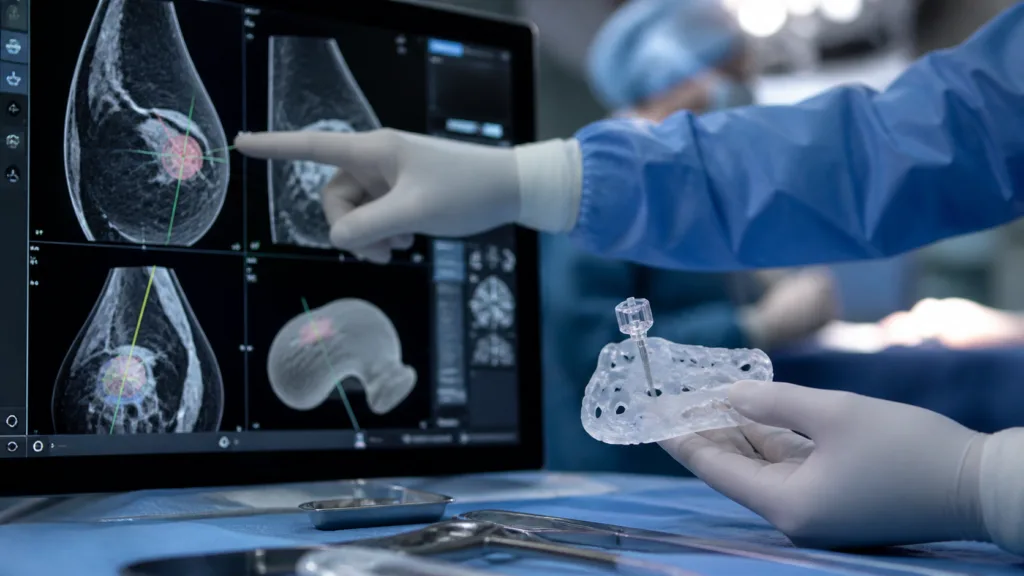
Wire localization has been used for decades because it gives surgeons a physical reference point for non-palpable lesions. Its weakness is that a single wire or limited reference pathway may not fully represent the three-dimensional shape of an irregular tumor. Cairn Surgical’s approach is different because the Breast Cancer Locator uses supine magnetic resonance imaging and a customized 3D-printed guide designed around the patient’s breast shape and tumor geometry.
That distinction matters because breast tumors are often not spherical, symmetrical, or easy to map using conventional localization tools. Cairn Surgical’s device attempts to give surgeons more patient-specific spatial information about tumor size, shape, location, and intended margin area while the patient is positioned for surgery. That could address a long-standing mismatch in breast surgery planning, where imaging may be acquired in one position while surgery is performed in another.
The risk is that more sophisticated localization may also introduce new dependencies. The Breast Cancer Locator requires supine magnetic resonance imaging at the patient’s hospital, image transfer to Cairn Surgical, device design, 3D printing, and delivery before surgery. That creates a more involved preoperative chain than wire localization alone. Hospitals may need to evaluate not only clinical performance but also whether turnaround times, imaging availability, production reliability, and patient scheduling can support broad use outside trial conditions.
Why trial design gives the data more weight but does not eliminate adoption uncertainty
The strength of Cairn Surgical’s pivotal trial lies in its randomized, prospective, multicenter design. The study enrolled 418 patients with non-palpable invasive breast cancer or ductal carcinoma in situ across 23 centers in the United States, Canada, the United Kingdom, and Austria. That structure gives the result more credibility than a single-center observational study because it reflects multiple surgeons, institutions, and care environments.
The inclusion of both invasive breast cancer and ductal carcinoma in situ is also relevant. Ductal carcinoma in situ and invasive lobular cancer are often associated with more challenging margin control, partly because disease extent can be harder to define. A device that improves localization across a mixed patient population could therefore have practical value for surgical teams managing complex non-palpable lesions.
Still, the headline reductions in positive margin rate and re-excision rate do not answer every regulatory or clinical question. Industry observers will want to see the full data set, including subgroup performance by tumor type, breast density, lesion size, imaging characteristics, surgeon experience, excised tissue volume, cosmetic outcomes, adverse events, and learning curve. A lower re-excision rate is powerful, but clinicians will also ask whether the device improves margin control without encouraging larger-than-necessary resections.
Why the technology’s real value may sit at the intersection of imaging, surgery, and customization
The Breast Cancer Locator is not merely a localization device in the traditional sense. It represents a broader trend in medical technology toward patient-specific procedural planning. By combining imaging data, 3D printing, and intraoperative physical guidance, Cairn Surgical is attempting to convert preoperative imaging into a surgical tool that can be used directly in the operating room.
That model is increasingly familiar in orthopedics, craniofacial surgery, and some interventional procedures, but breast cancer surgery has remained more dependent on conventional localization methods and surgeon judgment. If Cairn Surgical can show that a customized guide improves margin outcomes while preserving workflow reliability, the device could help shift breast surgery toward more individualized intraoperative navigation.
The limitation is that customization can be a double-edged sword. Patient-specific devices may offer better anatomical fit and procedural precision, but they can be harder to standardize, inventory, and reimburse than off-the-shelf tools. For medical device manufacturers, customization also raises questions about manufacturing quality systems, reproducibility, turnaround time, and scalability. Cairn Surgical will need to convince regulators, hospitals, and payers that the model is not only clinically elegant but operationally dependable.
What clinicians may watch next after the American Society of Breast Surgeons presentation
The presentation of the data at the American Society of Breast Surgeons annual meeting gives Cairn Surgical visibility among the clinicians most likely to influence adoption. Breast surgeons are practical evaluators. They will focus less on the novelty of 3D printing and more on whether the device improves decision-making before incision, reduces uncertainty during excision, and lowers the likelihood of a difficult conversation with patients after pathology results return.
Jennifer Gass, the trial’s principal investigator and Chief of Surgery at Women & Infants Hospital, indicated that the Breast Cancer Locator provided patient-specific information on tumor size, shape, and location based on supine magnetic resonance imaging, giving surgeons additional information to consider before and during surgery. That clinical framing is important because the device does not replace surgical judgment. It attempts to enhance the information environment in which that judgment is applied.
The challenge is that surgeons may differ in how they use that information. Some may see the Breast Cancer Locator as a precision tool for difficult cases, while others may want evidence supporting routine use in broader non-palpable breast cancer surgery. Adoption may therefore begin in specialized breast centers with access to magnetic resonance imaging, established multidisciplinary planning, and strong interest in reducing re-excision rates before expanding into more general hospital settings.
Why regulatory clearance will be the decisive commercial inflection point in the United States
Cairn Surgical stated that the Breast Cancer Locator remains investigational in the United States and is limited to investigational use. That makes the pivotal trial a major regulatory milestone rather than a commercial turning point by itself. The data may support a future marketing clearance pathway, but the final regulatory outcome will depend on the full submission package, device performance, safety profile, manufacturing controls, labeling, and the regulator’s assessment of clinical benefit.
For a device intended to guide breast tumor excision, regulators are likely to examine whether the clinical endpoint is meaningful, whether the comparator reflects standard practice, and whether the device can be used safely across varied surgical environments. The comparable safety profile reported in the trial is encouraging, but the regulatory question will be broader than whether adverse events were similar. It will include whether the device produces consistent guidance, whether the imaging-to-device workflow is robust, and whether user instructions adequately manage procedural variability.
Commercially, clearance would open the door to a more serious conversation with hospitals, ambulatory surgery centers, and breast cancer programs. However, reimbursement and cost justification may determine the pace of uptake. If the device can be linked to fewer re-excisions, lower downstream costs, improved patient experience, and better use of surgical capacity, the economic argument becomes stronger. If hospitals view it as an added procedural cost without clear payment support, adoption could be slower even with positive clinical data.
How Cairn Surgical fits into the broader shift toward precision-guided cancer surgery
Cairn Surgical’s pivotal trial sits within a larger movement in oncology toward precision not only in drugs and diagnostics but also in surgery. Breast cancer care has already been transformed by molecular subtyping, genomic testing, targeted therapy, and advanced imaging. Surgical localization has not moved as quickly, even though margin control remains central to breast-conserving treatment.
The Breast Cancer Locator brings that precision logic into the operating room by using patient-specific anatomy and imaging-derived tumor geometry to guide excision. If successful, it could strengthen the argument that surgical devices should be evaluated not merely by procedural convenience but by their ability to improve measurable oncologic and healthcare system outcomes.
The risk is that precision-guided surgery can sometimes overpromise if real-world adoption lags behind trial performance. Clinicians and hospital administrators will want evidence that the device works across diverse patient populations, does not create bottlenecks, and delivers value beyond specialized early adopters. The next phase for Cairn Surgical will therefore be as much about implementation evidence as pivotal trial evidence.
Why the next test is not only FDA review but hospital workflow acceptance
Cairn Surgical now has a clearer clinical story: its investigational Breast Cancer Locator reduced positive margins and re-excisions against wire localization in a randomized pivotal trial. That gives the medical device manufacturer a strong platform for regulatory engagement and clinical education. The bigger question is whether it can turn a promising trial result into a repeatable, scalable workflow for breast cancer surgery.
If the device secures U.S. clearance, early uptake is likely to depend on centers that already have the imaging infrastructure, surgical specialization, and operational discipline to support patient-specific device planning. Over time, broader adoption would require proof that the system can reduce avoidable second surgeries without placing too much strain on radiology scheduling, preoperative coordination, manufacturing logistics, or reimbursement pathways.
For breast surgeons, the appeal is straightforward: better tumor localization could mean cleaner margins, fewer repeat operations, and more confidence during difficult non-palpable excisions. For regulators and payers, the question is more demanding: whether the technology can deliver those outcomes consistently, safely, and economically. Cairn Surgical’s pivotal trial moves the Breast Cancer Locator closer to that conversation, but the commercial verdict will depend on how well precision guidance performs outside the controlled environment of a clinical study.