Pulse Biosciences, Inc. has begun enrolling patients in a first-in-human feasibility study evaluating the nPulse Vybrance Percutaneous Electrode System for treatment of T1N0M0 papillary thyroid microcarcinoma, a small and typically slow-growing form of thyroid cancer. The early clinical procedures have been completed at Sarasota Memorial Health Care System and The University of Texas MD Anderson Cancer Center as part of a prospective multi-center trial designed to evaluate safety and effectiveness of the nanosecond pulsed field ablation platform in tumors smaller than 1.5 centimeters.
The launch of the feasibility study marks an early but important step in determining whether nanosecond pulsed field ablation technology could open a new category of minimally invasive treatment options for early thyroid malignancies. While the announcement itself centers on patient enrollment, the broader implications relate to a long-standing clinical dilemma in thyroid oncology. Papillary thyroid microcarcinoma represents one of the most common thyroid cancers diagnosed globally, yet many cases progress slowly enough that clinicians must weigh intervention against overtreatment. Current management strategies typically revolve around either surgical thyroidectomy or active surveillance programs designed to monitor tumor growth. Both approaches carry limitations that have driven interest in less invasive alternatives capable of eliminating tumors without removing the thyroid gland.
Why the nonthermal mechanism of nsPFA technology could matter in anatomically delicate endocrine tumors
In this context, the Pulse Biosciences platform sits within a rapidly evolving field of energy-based tumor ablation technologies. The company’s system uses nanosecond pulsed field ablation, an electrical energy modality designed to disrupt cellular membranes while avoiding the thermal damage associated with conventional ablation methods such as radiofrequency or microwave energy. According to the company, the nPulse Vybrance system delivers extremely short electrical pulses that destroy targeted cells while preserving surrounding connective structures, including nerves and blood vessels.
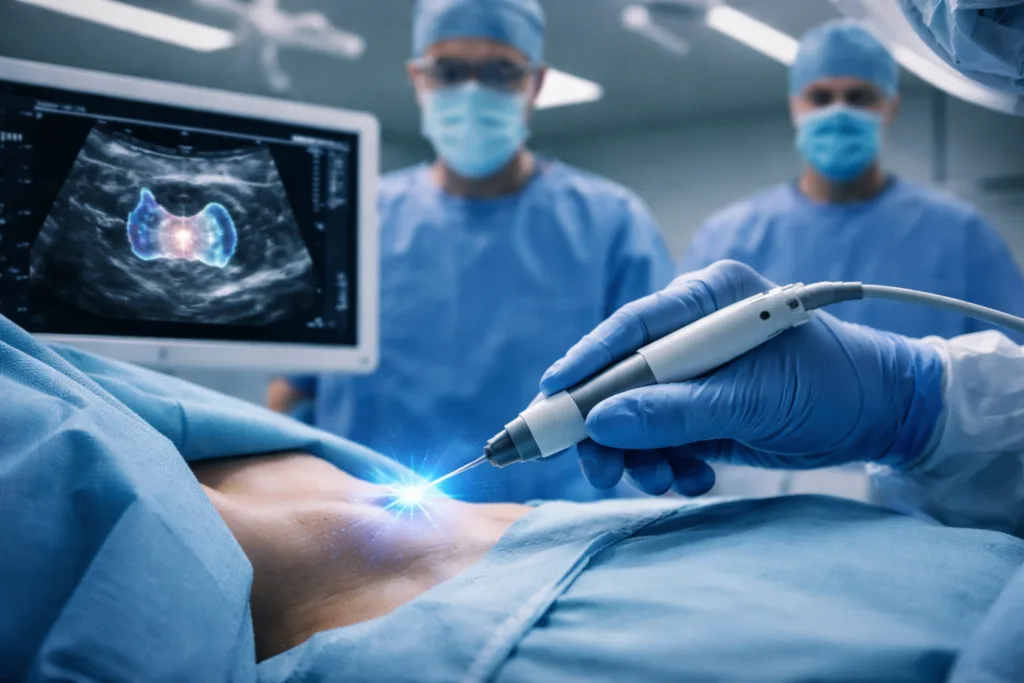
This mechanism of action has become a focal point of interest in recent years as clinicians explore ways to remove tumors located near delicate anatomical structures. In thyroid cancer treatment, protecting the recurrent laryngeal nerve and other nearby tissues is a critical surgical consideration. Even minor procedural damage can lead to complications such as vocal cord paralysis or long-term voice changes. The theoretical advantage of nonthermal ablation is that it may selectively target cellular tissue while sparing the structural scaffolding that surrounds it, potentially reducing the risk of these complications.
What the small feasibility study design reveals about early-stage regulatory and clinical strategy
The feasibility study itself remains small in scope, with enrollment planned for only thirty patients across two clinical sites. Such early trials are designed primarily to evaluate procedural safety and technical feasibility rather than clinical superiority. From a regulatory perspective, the device is being studied under an Investigational Device Exemption specifically for thyroid cancer use. The system has already received United States Food and Drug Administration 510(k) clearance for soft tissue ablation in other surgical settings, which may simplify the pathway for expanded indications if clinical evidence proves convincing.
Despite this regulatory foothold, the step from feasibility trial to routine clinical use remains substantial. Early device studies often demonstrate promising technical outcomes but face challenges when scaled into larger controlled trials. Observers tracking device innovation in oncology note that feasibility studies can reveal procedural practicality but rarely resolve questions about long-term recurrence rates, comparative effectiveness against surgery, or durability of tumor clearance. For papillary thyroid microcarcinoma specifically, the bar for demonstrating clinical value is unusually high because many patients already experience excellent outcomes with current treatment approaches.
Why papillary thyroid microcarcinoma presents a unique challenge for emerging interventional technologies
Active surveillance strategies have become increasingly accepted for small, low-risk thyroid cancers, particularly in Japan and more recently in the United States and Europe. Under this model, patients with tumors under a certain size may undergo periodic ultrasound monitoring rather than immediate surgery. Studies have shown that many such tumors remain stable for years without progression. As a result, any new intervention must demonstrate clear benefits either by improving quality of life, reducing anxiety associated with surveillance, or eliminating tumors with minimal procedural risk.
Energy-based ablation technologies have been explored before in this setting. Thermal methods such as radiofrequency ablation and laser ablation have been studied for benign thyroid nodules and, more cautiously, for small cancers. While these approaches can reduce tumor volume, they introduce the risk of heat diffusion damaging nearby structures. Nonthermal approaches such as nanosecond pulsed field ablation attempt to address this limitation by confining the destructive effect to cellular membranes rather than heating tissue.
What clinicians will evaluate when considering adoption of electrical field ablation platforms
Clinicians monitoring developments in the field suggest that the ultimate success of such technologies will depend not only on safety but also on procedural simplicity and reproducibility. If ablation techniques require complex imaging guidance or extended procedure times, adoption may remain limited to specialized centers. For thyroid tumors in particular, the ability to deliver precise treatment through a percutaneous needle electrode under ultrasound guidance could be a key determinant of clinical uptake.
The study design also highlights an emerging trend in oncology device trials involving collaboration with leading academic medical centers. Sarasota Memorial Health Care System and The University of Texas MD Anderson Cancer Center represent institutions with extensive experience in thyroid cancer management. Their participation in early clinical evaluation can accelerate data collection while lending credibility to emerging technologies. However, investigators in such trials typically treat carefully selected patients, meaning the results may not immediately translate to broader clinical populations.
Why long-term follow-up data will determine whether nsPFA becomes a credible clinical alternative
Another critical factor will be long-term follow-up data. Papillary thyroid microcarcinoma often progresses slowly, making short-term tumor destruction an incomplete measure of therapeutic success. Clinicians will want to see evidence that ablated tumors do not recur and that treated patients avoid delayed complications or secondary procedures. Achieving this level of evidence usually requires multi-year follow-up studies, which means meaningful conclusions about the technology’s effectiveness could take considerable time.
Industry observers also note that the commercial prospects of the platform extend beyond thyroid cancer alone. Pulse Biosciences has previously explored applications of its nanosecond pulsed field ablation technology in cardiac electrophysiology, particularly for treatment of atrial fibrillation. The underlying concept of electrical field ablation has already gained traction in cardiology through pulsed field ablation catheters developed by several major device manufacturers. Translating similar principles into oncology and surgical ablation represents a logical extension of the technology.
How Pulse Biosciences may position nsPFA as a broader surgical platform beyond thyroid cancer
From a strategic standpoint, the thyroid cancer feasibility study may therefore represent a proof-of-concept opportunity rather than a standalone product pathway. Demonstrating that nanosecond pulsed field ablation can safely eliminate tumors in delicate anatomical regions could encourage exploration in additional solid tumor settings. Potential future indications could include benign thyroid nodules, liver tumors, or other soft tissue lesions where precise tissue preservation is important.
However, several uncertainties remain. One major question is how consistently electrical field ablation can achieve complete tumor destruction in irregularly shaped lesions. Unlike surgical excision, which physically removes a tumor, ablation relies on energy distribution within tissue. Achieving uniform coverage across the entire tumor margin is technically challenging and may vary depending on tumor size, location, and electrode positioning.
What unresolved clinical, reimbursement, and scalability questions still surround the technology
Reimbursement considerations also represent a potential barrier to adoption. In healthcare systems where surgery is already well reimbursed and widely available, new device procedures must demonstrate cost effectiveness to gain coverage. Without clear economic advantages or patient outcome improvements, payers may hesitate to support widespread use.
Manufacturing scalability and training requirements also play roles in determining whether a new surgical device technology moves beyond experimental settings. Physicians must be trained to deliver the therapy safely, and healthcare systems must invest in compatible equipment such as energy consoles and specialized electrodes. These logistical factors often influence adoption as much as clinical evidence.
For now, the Pulse Biosciences feasibility trial should be viewed primarily as an exploratory milestone. Early patient treatments indicate that the procedure can be performed successfully in specialized centers, but the data needed to validate its clinical value will only emerge through continued enrollment and long-term follow-up.
Clinicians tracking the evolution of thyroid cancer management will be watching closely for signals about procedural safety, completeness of tumor ablation, and preservation of surrounding anatomical structures. Regulators will likely evaluate whether early outcomes justify larger pivotal trials that could support expanded device indications.
If the technology ultimately proves effective, it could offer a new middle ground between surgery and surveillance for small thyroid cancers. Such a shift would represent a broader trend toward minimally invasive tumor management using energy-based technologies that aim to treat disease while preserving organ function.