Soterix Medical Inc., a New Jersey-based manufacturer of non-invasive brain stimulation systems, has received U.S. Food and Drug Administration clearance for the SPRY Transcranial Magnetic Stimulation Therapy system, indicated for the treatment of major depressive disorder in adult patients who have not achieved satisfactory improvement from prior antidepressant medication in the current episode. The clearance marks Soterix Medical’s formal entry into the commercial TMS therapy market, a space already occupied by established incumbents including Neuronetics and BrainsWay, and positions the company to compete directly for the growing volume of treatment-resistant depression cases referred by outpatient psychiatry practices across the United States.
Why the SPRY clearance matters for a market that is still far from saturation in treatment-resistant depression care
The FDA clearance of SPRY TMS is not a scientific breakthrough in the conventional sense. Transcranial magnetic stimulation has been an FDA-cleared treatment for major depressive disorder since 2008, and the clinical evidence base underpinning it is well established. What the SPRY clearance represents instead is a commercial and design iteration on an existing standard of care, and those iterations, when executed well in a competitive device market, can have meaningful implications for how quickly TMS reaches clinical settings where adoption has so far been limited.
The TMS market in the United States remains underpenetrated relative to the scale of treatment-resistant depression as a clinical problem. Approximately 30 percent of patients with major depressive disorder fail to achieve adequate response from antidepressant pharmacotherapy, and that population represents tens of millions of Americans. Despite that unmet need, TMS adoption has been constrained by clinic-level friction rather than clinical doubt: the time burden of standard 36-session protocols, the workflow demands on clinical staff, the capital cost of device acquisition, and the ergonomic challenges of older systems have all slowed uptake. Soterix Medical’s value proposition with SPRY TMS appears to be targeted squarely at those adoption barriers, rather than at expanding the clinical indication itself.
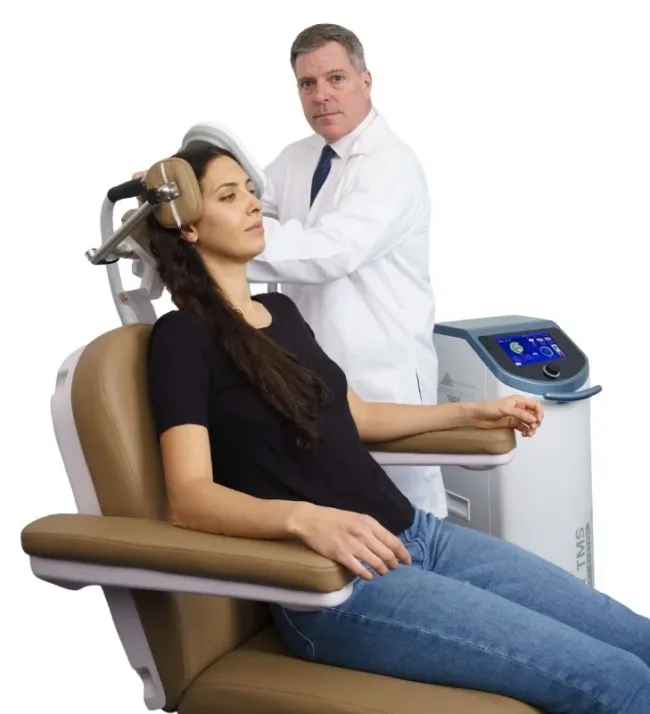
How SPRY TMS differentiates from existing cleared systems and what that means for clinical buyers
The SPRY system’s design features, as described by Soterix Medical, center on portability, ergonomics, and operational continuity. The integrated rapid liquid cooling system is a meaningful design distinction in a device category where heat management during continuous operation has historically required clinical interruptions. Standard TMS coils generate significant heat during treatment delivery, and systems without active cooling must pause to prevent equipment overheating, extending total session time and disrupting clinical scheduling. A cooling system that permits uninterrupted continuous operation, if validated in real-world clinical use, would represent a genuine workflow improvement for high-volume practices.
The on-device touchscreen interface and streamlined setup workflow reflect a broader trend in medical device design toward reducing the skill burden on clinical staff. TMS practices are often staffed by medical assistants or technicians rather than physicians for the actual treatment delivery phase, and system complexity at the device level creates a meaningful training and error-risk problem. Soterix Medical’s emphasis on a simplified setup pipeline suggests the company has studied where existing systems lose efficiency and has engineered accordingly.
The interchangeable coil design and compatibility with the SPRY POINT neuronavigation system adds a second tier of clinical functionality. Neuronavigation in TMS involves using imaging data to precisely target the dorsolateral prefrontal cortex, the primary stimulation site for depression treatment, rather than relying on standardized anatomical landmarks. For research-oriented practices and academic medical centers, neuronavigation compatibility positions SPRY TMS as a system that can serve both routine clinical and investigational applications from the same hardware platform, which has capital investment implications for buyers choosing between purpose-built clinical and research configurations.
What regulatory clearance via the 510(k) pathway reveals about the evidentiary standard Soterix Medical met
The FDA pathway for SPRY TMS clearance almost certainly proceeded through the 510(k) premarket notification process, which requires demonstration of substantial equivalence to a predicate device rather than independent clinical trial data. That pathway has governed TMS clearances in the United States since Neuronetics established the original predicate in 2008, and subsequent entrants including BrainsWay’s deep TMS system have navigated variants of that framework. Soterix Medical has not disclosed a specific clinical dataset supporting the SPRY clearance, which is consistent with 510(k) convention but leaves the clinical community without independent evidence on SPRY-specific response rates, remission outcomes, or tolerability profiles.
For clinicians making adoption decisions, the absence of published SPRY-specific clinical data is not disqualifying but it is a factor that will shape early uptake. The TMS indication itself carries a well-developed evidence base from the Neuronetics pivotal trials and subsequent real-world registry data, and a 510(k)-cleared system inherits the clinical credibility of that foundation. But practitioners selecting between competing cleared systems will be making purchase and referral decisions on operational, economic, and manufacturer-relationship criteria rather than comparative efficacy data, at least until SPRY-specific outcomes data is generated through post-market use.
Whether the partnership program structure accelerates or limits Soterix Medical’s market penetration rate
Soterix Medical has launched a formal partnership program for psychiatrists incorporating SPRY TMS, encompassing patient setup support, hands-on training and staff certification, financing options, and access to a referral network of other psychiatry practices. That commercial model reflects a sophisticated understanding of how TMS adoption actually unfolds at the practice level. The capital barrier for TMS entry is not trivial. Full-featured TMS systems are priced in the range of $60,000 to $150,000 depending on configuration, and the financing support component of Soterix Medical’s program is likely to be a more decisive factor for smaller and independent practices than any hardware feature distinction.
The referral network component is worth analytical attention. One of the structural inefficiencies in TMS adoption has been the challenge of building patient volume to a level that justifies the fixed overhead of maintaining a TMS-equipped practice. A manufacturer-facilitated network that connects practices and enables cross-referral has the potential to accelerate patient volume generation for early adopters in a way that organic referral development cannot. Industry observers note that this kind of commercial infrastructure support has become more common among device companies competing in the neuromodulation space, particularly as the market has matured enough to reward operational differentiation over product novelty.
What risks and unresolved questions could constrain SPRY TMS commercial performance over the next two years
Reimbursement remains the central structural risk for any new TMS entrant. The Centers for Medicare and Medicaid Services covers TMS for major depressive disorder, and most major commercial payers have coverage policies in place, but coverage parameters, prior authorization requirements, and session limits vary considerably across payers and states. Soterix Medical’s clearance does not resolve those reimbursement dynamics, and practices adopting SPRY TMS will encounter the same prior authorization friction and appeals burden that has limited the economics of TMS practice for other cleared systems.
The competitive positioning of SPRY TMS against Neuronetics’ NeuroStar system and BrainsWay’s deep TMS platform will depend heavily on Soterix Medical’s ability to execute on its service and support infrastructure at scale. Both incumbents have built substantial post-sale support ecosystems, proprietary clinical protocols, and patient access programs over more than a decade of market presence. A new entrant with a differentiated hardware platform but a thinner support ecosystem faces a meaningful credibility gap in procurement conversations with health systems and large group practices, where capital committee scrutiny of vendor stability is a standard gatekeeping mechanism.
The portability and ergonomic design claims attached to SPRY TMS will also face the discipline of real-world clinical validation over the months following commercial launch. Design advantages demonstrated in controlled demonstrations do not always survive the complexity of high-volume outpatient settings, where patient throughput, staffing variability, and equipment maintenance realities test every claim of operational simplicity. Regulatory watchers and clinical early adopters will be watching post-market performance data carefully, particularly on the continuous operation and cooling system claims that represent the most differentiated technical assertions Soterix Medical has advanced for SPRY.
A final unresolved question is whether Soterix Medical intends to pursue additional indications beyond major depressive disorder. The FDA has cleared TMS for obsessive-compulsive disorder and smoking cessation in addition to depression, and the deep TMS platform from BrainsWay has pursued a broader indication strategy. Whether SPRY TMS is positioned as a depression-focused product or as a platform intended to expand across neuromodulation indications will have significant implications for the long-term commercial trajectory of the system and for how Soterix Medical allocates its clinical and regulatory investment in the years ahead.