Why Rapid Medical’s DISTALS data could reshape treatment expectations in smaller stroke vessels
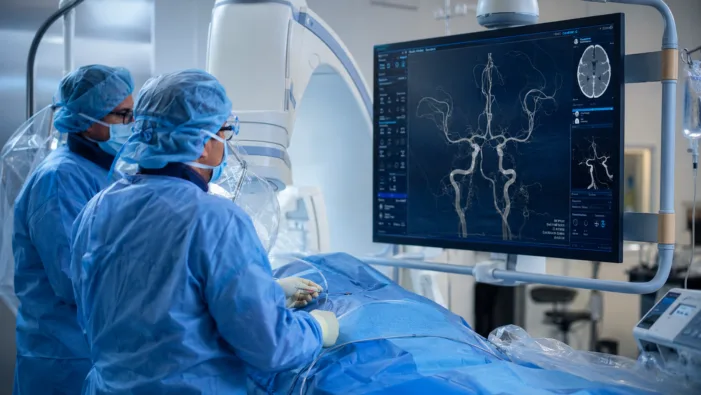
Distal stroke has long lacked device options. Rapid Medical’s TIGERTRIEVER 13 data now tests whether small-vessel thrombectomy can scale.

Distal stroke has long lacked device options. Rapid Medical’s TIGERTRIEVER 13 data now tests whether small-vessel thrombectomy can scale.
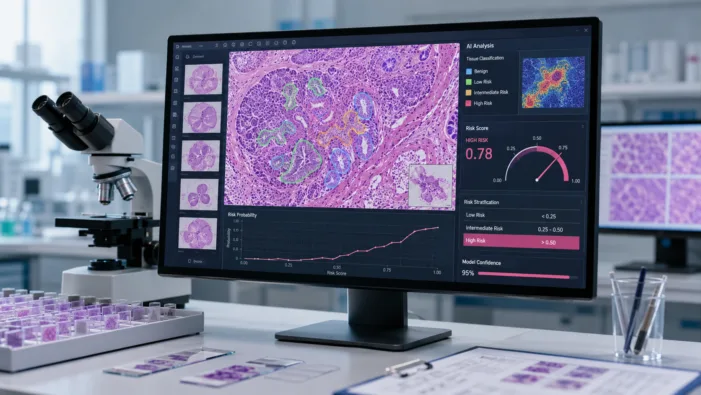
AI can read pathology slides, but can it change breast cancer treatment decisions? ArteraAI Breast puts that question before clinicians and payers.
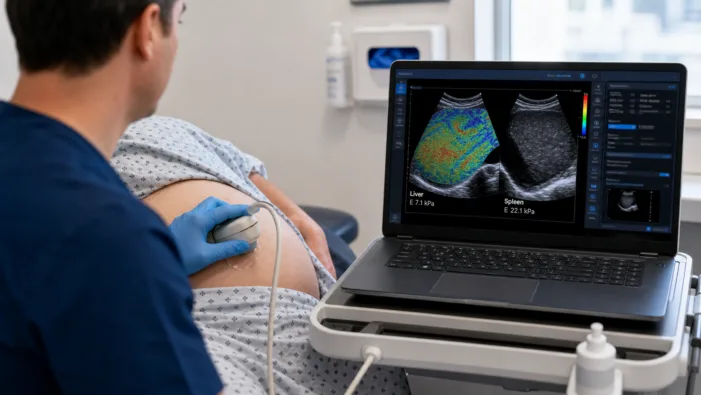
Liver diagnostics need speed and scale. E-Scopics’ Hepatoscope clearance tests whether bedside elastography can move deeper into routine care.

Pediatric ear care has long had a waiting-room problem. Earflo’s FDA-cleared device tests whether home therapy can close the gap.

CorVista Health presents machine-learned non-invasive INOCA detection data at ACC.26. Analysis of what this means for cardiology and women’s heart care. Read more.
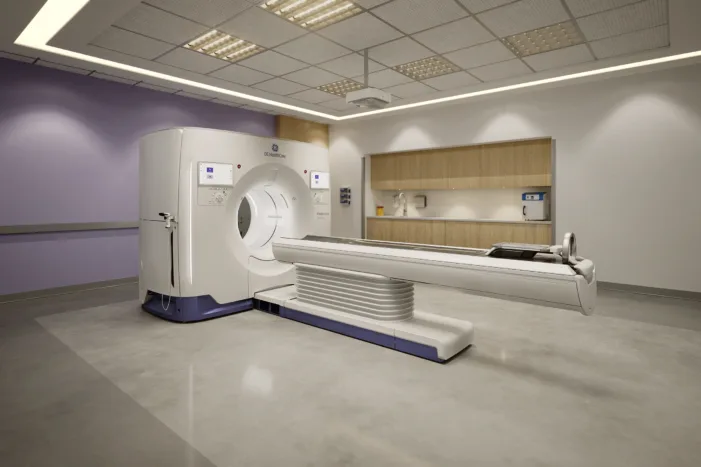
GE HealthCare’s Photonova Spectra just won FDA clearance. Find out what this means for photon-counting CT, imaging workflows, and hospital adoption.
QIAGEN wins FDA clearance for GI panels on QIAstat-Dx Rise. Discover what this means for syndromic testing platforms and molecular diagnostics adoption.
Soterix Medical wins FDA clearance for SPRY TMS in treatment-resistant major depression. What the clearance means for the neuromodulation market. Read the analysis.

GE HealthCare secures FDA clearance for next-generation SIGNA MRI. Explore clinical impact, workflow gains, and market implications.

Medtronic wins FDA clearance for Stealth AXiS in spine robotics. Explore what this integrated platform means for workflow, data, and adoption.