HAI Solutions has received De Novo classification from the U.S. Food and Drug Administration (FDA) for its QIKCAP System, making it the first ultraviolet light-based microbial reduction device to be granted such approval for use with needleless luer-activated IV connectors. The FDA decision establishes a new Class II medical device category, placing the QIKCAP platform in uncharted regulatory territory for ultraviolet microbial disinfection tools used in critical care IV therapy.
The decision marks a meaningful inflection point for infection control within high-risk hospital environments such as ICUs, operating rooms, and emergency departments, where central line-associated bloodstream infections (CLABSIs) remain a costly and persistent threat. With the QIKCAP system, HAI Solutions is now positioned to lead a niche but increasingly strategic segment of the market where UVC light is being integrated into catheter maintenance protocols.
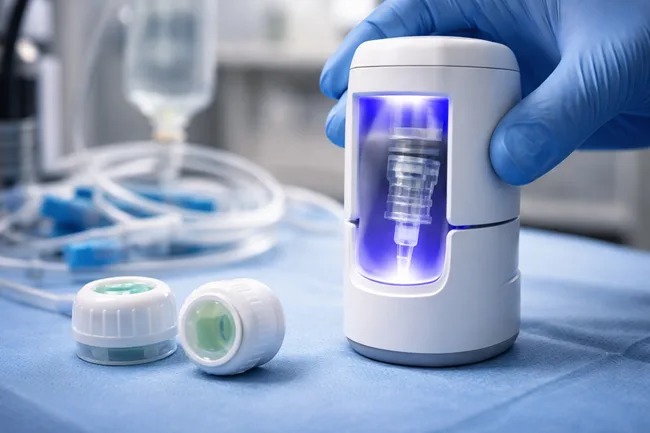
What the FDA De Novo grant signals about the agency’s evolving stance on UVC devices
The De Novo grant for the QIKCAP system carries broader implications than its initial regulatory footprint might suggest. While ultraviolet C (UVC) irradiation has been investigated in various hospital settings—including surface disinfection and air purification—FDA clearance for direct application on vascular access devices has remained elusive due to concerns around reproducibility, patient safety, and clinical outcome correlation.
By formally creating a new Class II category for UVC microbial reduction on luer-activated valves, the FDA is signaling a measured yet significant shift in its stance. Regulatory observers note that the agency’s decision to establish general and special controls specifically for this technology suggests it may now be more receptive to high-efficacy UVC interventions with clearly defined use protocols, dosing standards, and technical validations.
HAI Solutions reportedly worked closely with the agency through the De Novo process, submitting performance testing data under worst-case simulated conditions, including high levels of microbial contamination and incomplete manual disinfection. The device demonstrated strong efficacy—achieving 4-log (99.99%) reductions in common hospital-acquired pathogens such as Staphylococcus aureus, Klebsiella pneumoniae, Staphylococcus epidermidis, and Enterobacter cloacae—when used in conjunction with CHG/IPA wipes.
However, it fell short on fungal organisms and has yet to show a direct clinical correlation with reduced infection outcomes, a point that infection prevention specialists are expected to monitor closely in post-market adoption.
Why QIKCAP’s mechanism matters in the context of IV connector disinfection challenges
Clinicians familiar with central line protocols know that needle-free connectors—while reducing needlestick injuries—also present unique microbial risks. These devices have a known vulnerability: the septum, or access membrane, which can harbor pathogens despite standard wipe-based disinfection. This has led to inconsistent efficacy depending on scrub time, technique, and compliance, especially in high-pressure or time-sensitive care settings.
HAI Solutions’ QIKCAP platform introduces a two-part defense: a 10-second UVC irradiation step followed by application of a sterile cap that acts as a physical barrier for up to seven days. This dual-action approach attempts to remove user variability from the disinfection equation while adding a passive layer of contamination prevention.
While this mechanism is not entirely novel in theory—UVC has long been known to deactivate bacterial DNA—the regulatory breakthrough lies in the device’s successful alignment with FDA controls for safety and efficacy in a narrow clinical use case. For healthcare institutions seeking to reduce bloodstream infections without fully overhauling central line workflows, the QIKCAP system could offer a modular and scalable solution.
What adoption hurdles still remain despite regulatory greenlighting
Despite gaining De Novo classification, QIKCAP will face hurdles that extend beyond regulatory approval. Chief among them is the gap between microbial reduction in bench testing and demonstrated clinical impact in real-world use. Infection prevention programs and hospital procurement teams are increasingly under pressure to justify new device costs based on patient outcome improvements or cost savings from reduced complications.
Without data linking QIKCAP use to reductions in CLABSI rates or associated hospital stay durations, administrators may hesitate to displace existing manual disinfection protocols that are cheaper and more broadly validated.
Moreover, there is a question of workflow integration. The need to perform a UVC step before capping, although fast, still requires a behavioral shift for nurses and care staff. The 10-second cycle may be efficient in isolation, but could become a bottleneck in high-volume units unless implementation is supported with sufficient devices and staff training.
Industry observers also note that the product’s fungal coverage limitations—along with potential battery performance or cap shelf life constraints—could impact confidence and restrict early use to controlled settings rather than widespread rollout.
Where QIKCAP fits within the competitive and technological landscape of catheter care
The QIKCAP approval may catalyze a new product category within the infection prevention device market, but it will need to compete with both entrenched protocols and emerging rivals. Established products like antimicrobial-impregnated catheter caps, disinfecting port protectors, and CHG dressings have years of data behind them, along with institutional trust and volume-based procurement advantages.
That said, ultraviolet disinfection has a growing foothold in other hospital technologies, such as UVC robots and air-handling systems. HAI Solutions’ ability to position QIKCAP as part of a broader, hospital-wide UVC strategy may help it win early adoption among infection control teams already familiar with light-based microbial reduction.
The company’s partnership with Intellego Technologies for a chromatic UVC dosimeter may also help build trust, offering visual confirmation that sufficient irradiation has occurred—an important usability feature in healthcare environments where over- or under-treatment risks are serious.
If HAI Solutions can couple its regulatory win with a robust clinical study pipeline and effective real-world implementation support, it may shape a new frontier in catheter-related infection prevention—one that bridges the gap between manual disinfection and fully passive protection.
What regulators, clinicians, and competitors are likely to watch next
The next chapter in QIKCAP’s trajectory will likely be shaped by post-market surveillance and peer-reviewed clinical research. Infection control committees will want data from controlled deployments comparing infection incidence, protocol compliance, and economic impact across patient populations.
Meanwhile, other device manufacturers exploring UVC for vascular access or catheter site protection may view this De Novo clearance as a beachhead. Several companies have shelved similar programs due to regulatory uncertainty, but the newly created device classification may re-open the field for innovation.
Clinicians will also monitor patient safety signals, particularly around unintended consequences such as skin exposure risks, cap detachment, or potential impact on luer valve material integrity over time. If early adopters find the technology reliable and easy to use, QIKCAP could influence future procurement cycles.
For now, HAI Solutions has crossed a key regulatory hurdle. The true test will come in whether QIKCAP can establish not just microbial efficacy, but meaningful clinical value.