HAI Solutions earns FDA De Novo clearance for QIKCAP UVC IV disinfection device
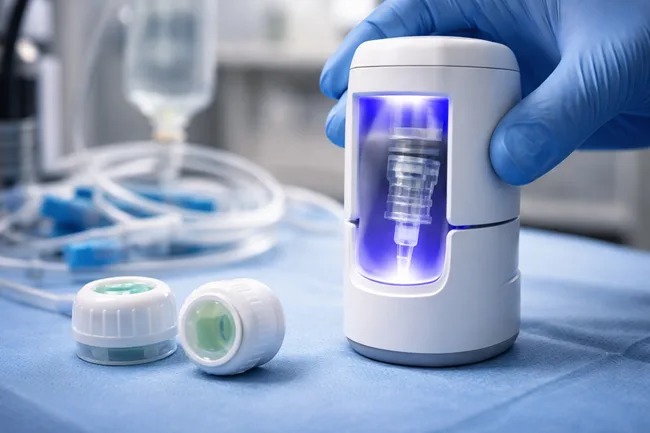
HAI Solutions has received De Novo classification from the U.S. Food and Drug Administration (FDA) for its QIKCAP System, making it the first ultraviolet light-based microbial reduction device to be granted such approval for use with needleless luer-activated IV connectors. The FDA decision establishes a new Class II medical device category, placing the QIKCAP platform […]