Johnson & Johnson has introduced its VARIPULSE pulsed field ablation system in Peru for the treatment of drug-refractory paroxysmal atrial fibrillation, marking the arrival of the company’s next generation electrophysiology technology in the Latin American market. The procedure has been performed at Clínica Delgado, part of the AUNA healthcare group, where physicians are evaluating the integrated mapping and ablation platform for patients whose atrial fibrillation cannot be controlled through pharmacological therapy.
How pulsed field ablation is emerging as a potential alternative to traditional thermal ablation technologies
The introduction of pulsed field ablation in Peru reflects a broader technological shift underway in the treatment of atrial fibrillation. For more than two decades, catheter ablation procedures have primarily relied on thermal energy sources such as radiofrequency or cryothermal ablation. These methods destroy cardiac tissue responsible for abnormal electrical signaling through heat or freezing.
Pulsed field ablation introduces a fundamentally different mechanism. Instead of thermal injury, the technology delivers controlled electrical pulses that induce irreversible electroporation in cardiac cells. The process creates microscopic pores in cell membranes, leading to cell death in targeted myocardial tissue while potentially sparing adjacent anatomical structures.
This tissue selectivity has become one of the main drivers behind industry interest in pulsed field ablation. Thermal ablation techniques can occasionally damage nearby tissues including the esophagus, pulmonary veins, or phrenic nerve. Although such complications are relatively rare, they remain among the most serious risks associated with atrial fibrillation ablation procedures.
Industry observers tracking electrophysiology innovation suggest that pulsed field ablation could reduce some of these risks by targeting myocardial tissue more selectively. If confirmed through large clinical datasets, this property could reshape the safety profile of atrial fibrillation ablation procedures and potentially broaden their use.
Why integrated mapping and ablation platforms could change electrophysiology procedural efficiency
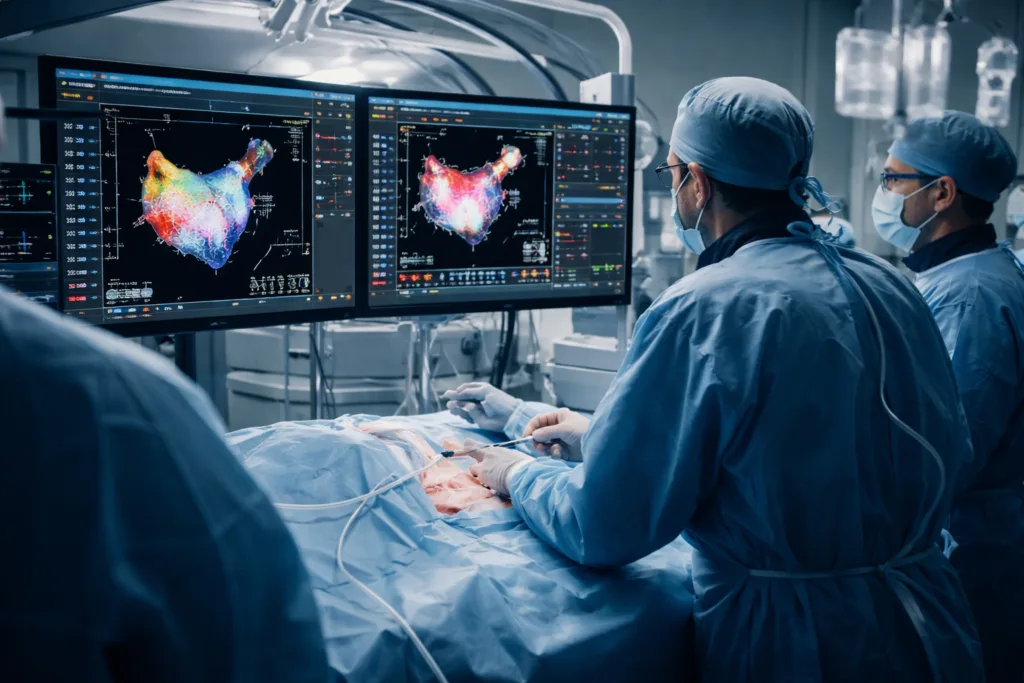
Johnson & Johnson’s VARIPULSE platform is built around an integrated system that combines cardiac mapping and ablation within a single procedural environment. Electrophysiologists rely on three dimensional mapping systems to identify abnormal electrical pathways inside the heart and guide catheter positioning during ablation procedures.
Traditionally, mapping systems and ablation catheters operate through separate technologies that must be coordinated during the procedure. This can introduce workflow complexity, particularly in high volume electrophysiology laboratories performing large numbers of ablation procedures each year.
The integrated design of the VARIPULSE platform aims to streamline this process. Real time visualization of cardiac structures and electrical activity allows electrophysiologists to map arrhythmogenic regions and deliver pulsed field energy within the same procedural interface.
Clinicians involved in early evaluations of the technology indicate that this integration could shorten procedural times and reduce fluoroscopy exposure. Lower fluoroscopy use is particularly important in electrophysiology because physicians performing catheter based interventions are exposed to radiation repeatedly over the course of their careers.
Reduced procedural complexity may also improve reproducibility across electrophysiology laboratories. In many hospitals, atrial fibrillation ablation outcomes can vary depending on operator experience and institutional workflow. Technologies that simplify procedural steps may help standardize results across different healthcare systems.
What Johnson & Johnson’s Peru launch reveals about the global expansion of pulsed field ablation
The deployment of pulsed field ablation technology in Peru highlights how rapidly the technology is expanding beyond early adopter markets. Electrophysiology centers in the United States and Europe were among the first to begin evaluating pulsed field ablation platforms as clinical trial results emerged in recent years.
Device manufacturers are now moving to expand access into emerging markets where cardiovascular disease remains a major health burden. Latin America represents one such region, where aging populations and rising rates of cardiovascular risk factors are contributing to increasing atrial fibrillation prevalence.
Atrial fibrillation is currently estimated to affect more than 50 million people worldwide. The condition significantly increases the risk of stroke, heart failure, and hospitalization. When medications fail to maintain rhythm control, catheter ablation becomes one of the most effective treatment options available.
Expanding access to advanced ablation technologies could therefore have important implications for regional cardiac care. Hospitals capable of performing modern electrophysiology procedures can reduce dependence on international referral centers while improving treatment availability for local patients.
For Johnson & Johnson, the launch also reflects a strategic effort to strengthen its cardiovascular device portfolio within the company’s MedTech division. The healthcare conglomerate has historically maintained a strong presence in electrophysiology through mapping systems and catheter technologies, but the emergence of pulsed field ablation has intensified competition among device manufacturers.
Several companies across the cardiac rhythm management sector are now developing their own pulsed field ablation platforms, turning the technology into one of the fastest evolving segments within cardiovascular medical devices.
Why long term clinical durability and real world outcomes will determine the success of pulsed field ablation
Despite growing enthusiasm around pulsed field ablation, several clinical questions remain unresolved. One of the most important issues involves lesion durability, which determines whether ablation procedures deliver lasting rhythm control.
Successful atrial fibrillation ablation requires permanent electrical isolation of the pulmonary veins, preventing abnormal electrical signals from entering the left atrium. If the lesions created during ablation do not remain durable over time, patients may experience recurrence of arrhythmia and require repeat procedures.
Thermal ablation technologies have accumulated more than two decades of clinical experience demonstrating durable outcomes when performed correctly. Pulsed field ablation systems are still building that long term evidence base.
Regulatory watchers therefore expect that multicenter trials and real world registries will play a major role in determining whether pulsed field ablation platforms achieve comparable durability. The ability to demonstrate sustained arrhythmia control will ultimately influence how widely physicians adopt the technology.
What adoption barriers and economic considerations could influence pulsed field ablation rollout
Beyond clinical evidence, several operational factors will influence how quickly pulsed field ablation systems spread across healthcare systems. Electrophysiology laboratories must adapt to new catheter designs, energy delivery systems, and procedural workflows.
Training requirements can slow early adoption, particularly in hospitals where electrophysiology teams are already accustomed to thermal ablation techniques. Physicians must become familiar with new mapping strategies, catheter manipulation techniques, and lesion assessment methods.
Reimbursement policies also play a significant role in technology adoption. Hospitals often evaluate whether new procedural platforms provide measurable benefits such as shorter procedure times, lower complication rates, or reduced hospitalization costs.
In emerging markets, cost effectiveness considerations may become even more important. Healthcare systems with limited budgets must weigh the value of advanced technologies against competing priorities for medical investment.
Manufacturing capacity and supply chain logistics represent additional considerations as pulsed field ablation expands globally. Device manufacturers must ensure that catheter production and system deployment can scale efficiently across different geographic markets.
Why the arrival of pulsed field ablation in Peru signals a broader shift in electrophysiology innovation cycles
The introduction of the VARIPULSE system in Peru illustrates how the diffusion of medical technology is accelerating across global healthcare markets. Historically, advanced electrophysiology technologies often appeared first in large academic centers in North America or Europe before reaching other regions years later.
The relatively rapid expansion of pulsed field ablation into Latin America suggests that innovation cycles in cardiovascular medicine are shortening. As global healthcare networks become more interconnected, hospitals in emerging markets are increasingly able to adopt new technologies closer to their initial commercial rollout.
For physicians and healthcare systems in Peru, the availability of pulsed field ablation may represent an opportunity to participate in the next phase of electrophysiology innovation. As clinicians begin evaluating the technology in real world practice, the data generated from these procedures will contribute to the growing global evidence base surrounding pulsed field ablation.
Ultimately, the long term success of pulsed field ablation will depend on whether clinical evidence confirms the safety and procedural advantages that early studies have suggested. Cardiologists, regulators, and medical device manufacturers will be watching closely as hospitals worldwide accumulate experience with this new generation of atrial fibrillation treatment technology.