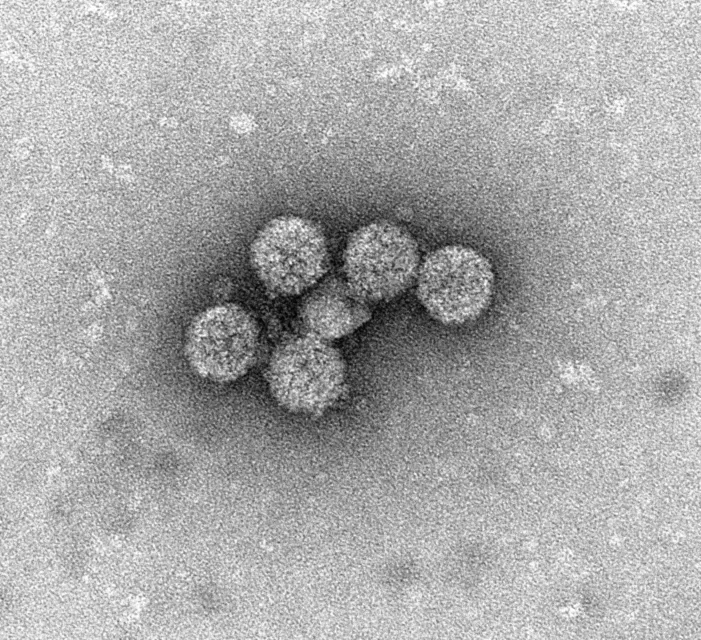
Could Hyundai Bioscience’s XAFTY become a crisis-response antiviral for Ebola and hantavirus?

Ebola urgency is rising, but broad-spectrum antivirals still face a proof gap. Hyundai Bioscience’s XAFTY now enters that debate.

Ebola urgency is rising, but broad-spectrum antivirals still face a proof gap. Hyundai Bioscience’s XAFTY now enters that debate.
Orthogon Therapeutics raised $11M more for its BK virus drug program. Read why this transplant antiviral story could matter far beyond niche biotech.

ArkBio has begun Phase I testing of AK0406 in Australia. Read why this long-acting influenza candidate matters and what risks still need proving.

Island Pharmaceuticals Limited has confirmed that the United States Food and Drug Administration (FDA) continues to evaluate its most recent regulatory submission related to Galidesivir, the company’s clinical-stage antiviral candidate being advanced under the Animal Rule pathway. In correspondence dated 2 January 2026, the regulator informed the Australian biotech firm that additional time is required […]