Why CUE-401’s IMMUNOLOGY2026 data could matter for autoimmune drug pipelines

Cue Biopharma, Inc. unveils new CUE-401 data. Find out what this means for autoimmune therapy development and the regulatory path ahead.

Cue Biopharma, Inc. unveils new CUE-401 data. Find out what this means for autoimmune therapy development and the regulatory path ahead.

Hinge Bio, Inc. doses first subjects in HB2198 Phase 1 trial. Discover what this could mean for lupus treatment and next-generation immune reset therapies.

Read why Merakris Therapeutics’ MTX-001 data may sharpen industry focus on refractory wound healing and reshape the late-stage wound care outlook.
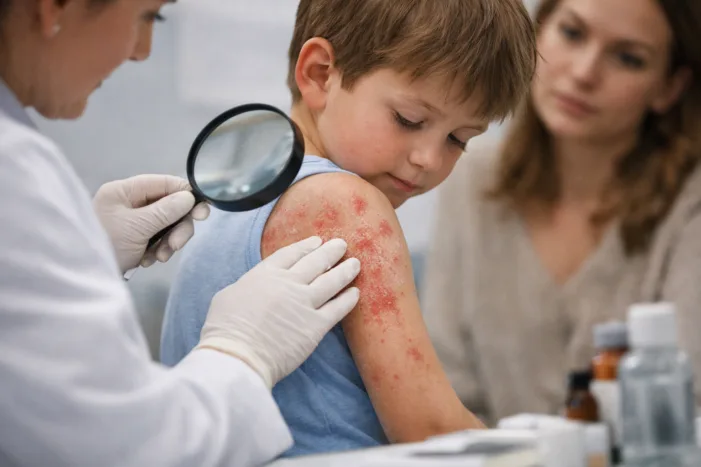
Galderma’s nemolizumab phase II data in children aged 2-11 show 52-week itch and lesion relief. Read the analysis.

Apogee Therapeutics prepares to release 52-week APEX data for zumilokibart. Discover what it could mean for eczema treatment and competition.
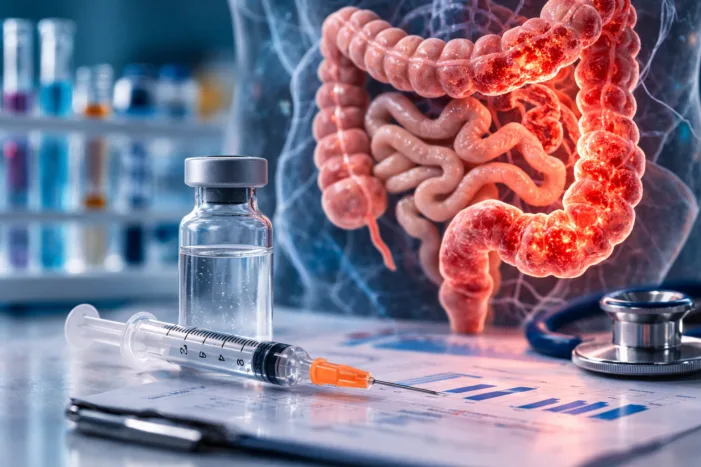
AbbVie reports positive Phase 3 AFFIRM trial results for risankizumab subcutaneous induction in Crohn’s disease. Discover what the findings mean for treatment.
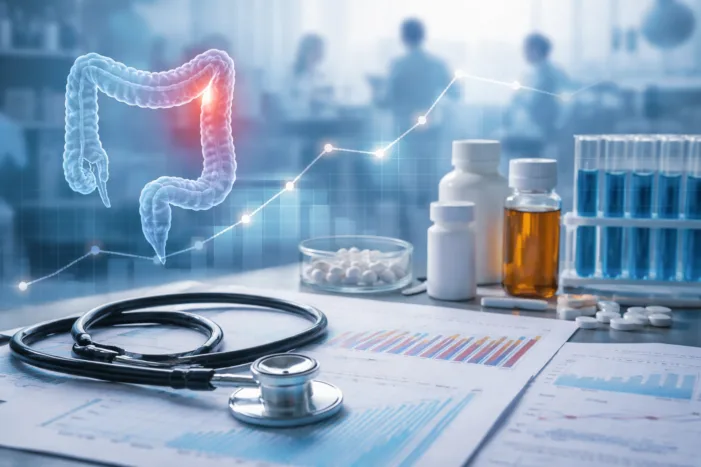
Johnson & Johnson’s three-year TREMFYA data highlight why sustained remission is redefining ulcerative colitis treatment decisions. Read the analysis.

Could a nasal spray replace flu shots? Find out how Leyden Labs’ CR9114 antibody is changing the game in influenza prevention.

Find out why Boehringer is betting big on Simcere’s bispecific SIM0709 to challenge the IBD treatment ceiling.

Veloxis earns FDA orphan status for pegrizeprument in liver transplant rejection. Find out what this means for the future of immunosuppressive therapy.