Is Amgen’s bispecific lung cancer drug becoming a global standard after chemotherapy failure?
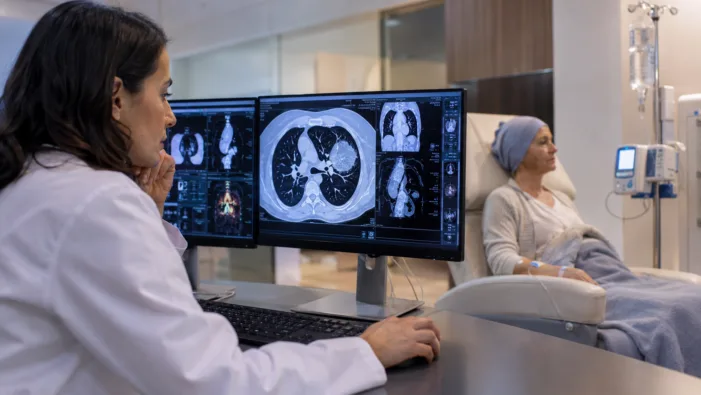
Amgen’s Imdylltra has European approval in relapsed small cell lung cancer. Survival data now face reimbursement and adoption tests.

Amgen’s Imdylltra has European approval in relapsed small cell lung cancer. Survival data now face reimbursement and adoption tests.
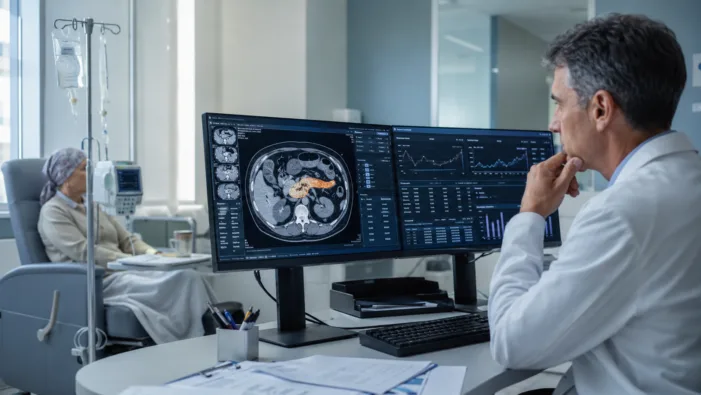
See how Phanes Therapeutics’ spevatamig data could reshape metastatic pancreatic cancer drug development after ASCO 2026.

Rare cancer biology met regulatory urgency. Bizengri’s approval tests whether precision oncology can reach tiny patient subsets fast.
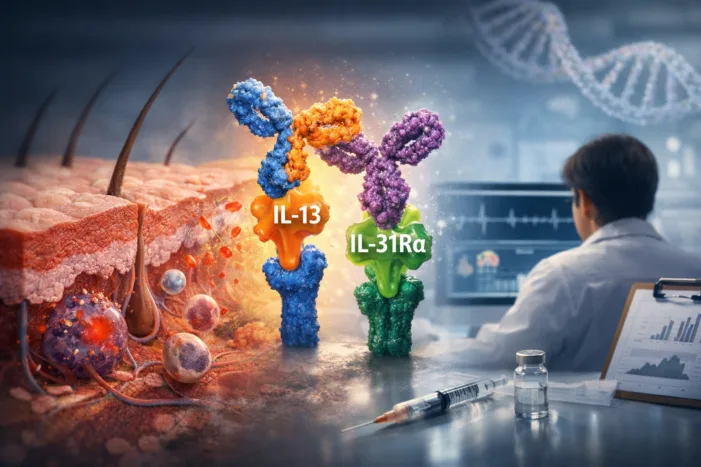
Zai Lab’s ZL-1503 showed durable preclinical itch and inflammation control. Read what its 2026 clinical path could mean for atopic dermatitis.

Bambusa Therapeutics’ BBT001 showed strong Phase 1 activity in atopic dermatitis. Read what it could change and what risks remain.

NEOK002 has cleared the FDA IND stage. Read why this bispecific ADC matters for solid tumors, clinical strategy, and the next phase of ADC competition.

Gilead Sciences acquires Ouro Medicines for up to $2.18bn, adding BCMA T cell engager gamgertamig to its inflammation pipeline. Analysis of what the deal reveals.

MindWalk launches B Cell Llama, a VHH nanobody platform for bispecific drug design. Peer-reviewed data raises key questions about function, immunogenicity, and AI.

FDA approves J&J’s teclistamab plus daratumumab for relapsed myeloma from second line. Analyse the MajesTEC-3 data, safety profile, and what this changes for treatment.

TECVAYLI outperformed standard regimens in relapsed/refractory myeloma. Find out what the Phase 3 MajesTEC-9 trial means for future treatment lines.