Can REYOBIQ improve Plus Therapeutics’ path in pediatric high-grade glioma and ependymoma?
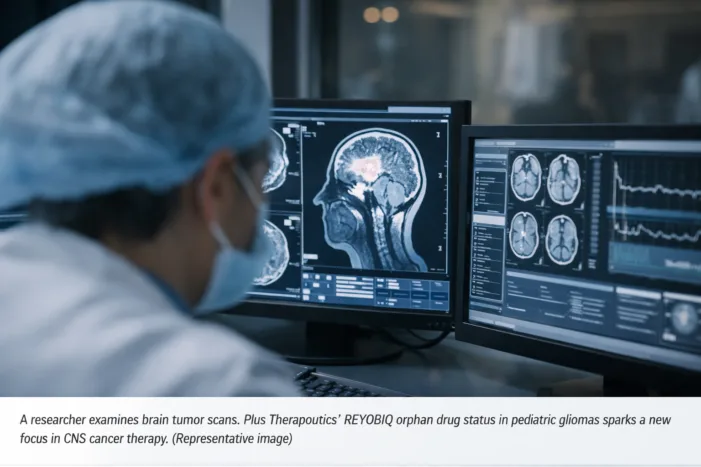
Plus Therapeutics won orphan drug designation for REYOBIQ in pediatric malignant gliomas. Read what it changes for CNS cancer development.

Plus Therapeutics won orphan drug designation for REYOBIQ in pediatric malignant gliomas. Read what it changes for CNS cancer development.

Find out how Adaptin Bio is preparing a Phase 1 glioblastoma trial and why its preclinical progress matters for brain cancer treatment innovation.

Curasight A/S has dosed the first patient in a Phase 1 clinical trial of uTREAT for glioblastoma, launching the therapeutic half of its uPAR-targeting theranostic platform alongside its companion diagnostic, uTRACE, which is already in Phase 2 for prostate cancer. How curative ambitions shift when a theranostic platform enters human testing in glioblastoma Curasight’s advancement […]

T-MAXIMUM’s MT027 CAR-T therapy for glioblastoma enters Phase II after FDA clearance. Find out what this means for solid tumor cell therapy.