Can enicepatide and petrelintide help Genentech build a differentiated obesity franchise?

Genentech advances enicepatide and petrelintide obesity data at ADA 2026. Find out why Roche’s strategy matters now.

Genentech advances enicepatide and petrelintide obesity data at ADA 2026. Find out why Roche’s strategy matters now.
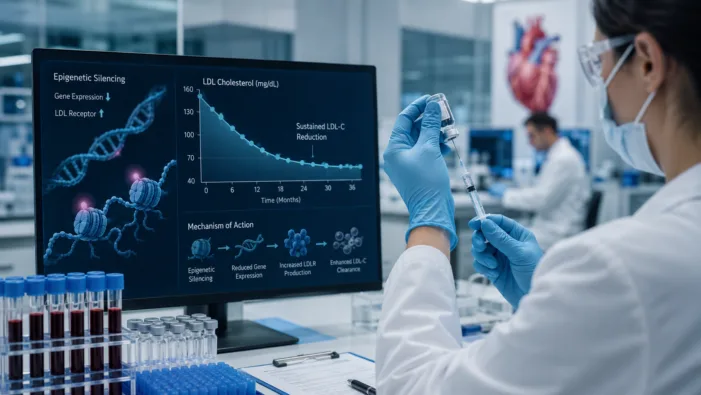
Find out how Scribe Therapeutics’ STX-1150 could reshape long-acting LDL-C lowering through CRISPR-based epigenetic silencing.

CRISPR can lower cholesterol. The harder test is whether Scribe can make durability, precision, and scalability work in common disease.

GLP-1 drugs dominate obesity. GSK is betting SiranBio’s SA030 can attack visceral fat risk from a different angle.

MetaVia has started higher-dose testing for DA-1726 in obesity. Read what this means for tolerability, competition, and the next clinical milestone.

Merck’s enlicitide decanoate beat oral rivals in Phase 3. Read what this could change for cholesterol care, PCSK9 adoption, and market strategy.

Can Merck turn WINREVAIR’s CADENCE data into a new treatment path in CpcPH-HFpEF? Read the full analysis of what changes next.

Scribe Therapeutics hits a second in vivo CRISPR milestone with Eli Lilly. Explore what this changes for gene editing platforms and future therapies.