Incyte’s frontMIND data put pressure on decades-old R-CHOP dominance in high-risk DLBCL
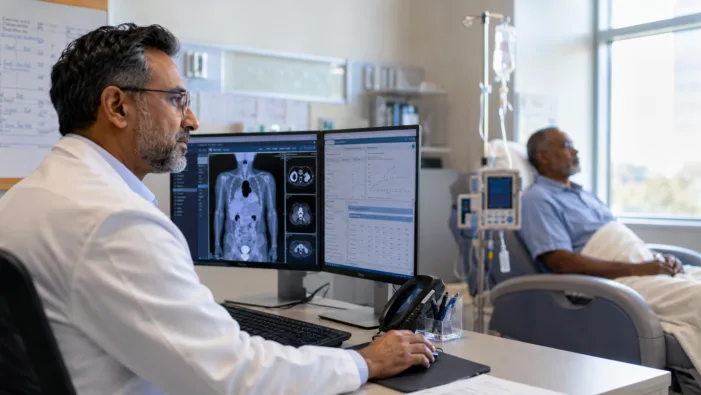
Find out how Incyte’s tafasitamab data could reshape first-line treatment expectations in high-risk DLBCL.

Find out how Incyte’s tafasitamab data could reshape first-line treatment expectations in high-risk DLBCL.

CAR-T is powerful but hard to scale. Therorna’s circRNA platform tests whether in vivo engineering can move the field beyond bespoke therapy.

Can CTA313 redefine lupus treatment with CAR-T? Explore what Imviva Biotech’s early data means for autoimmune therapy innovation.

Tempest Therapeutics, Inc. advances TPST-2003 CAR-T data. Discover how dual-targeting could impact multiple myeloma treatment durability.

Find out how Tempest Therapeutics, Inc.’s CAR-T strategy could shape the next phase of cell therapy competition and multiple myeloma innovation.

Hinge Bio, Inc. doses first subjects in HB2198 Phase 1 trial. Discover what this could mean for lupus treatment and next-generation immune reset therapies.

Innovent Biologics unveiled preclinical IBI3055 data in autoimmune disease. Read what this could change for immune-reset therapies.

AvenCell Therapeutics has dosed the first AVC-203 patient in QUADvance. Read what this means for allogeneic CAR-T and B-cell cancer treatment.

Eli Lilly and Company has entered into a definitive agreement to acquire Orna Therapeutics, Inc., gaining access to the biotechnology firm’s circular RNA platform and its clinical trial ready in vivo CAR-T candidate ORN-252 targeting CD19 for B cell driven autoimmune diseases. The deal, valued at up to 2.4 billion dollars in cash including milestone […]