Can AeroRx turn nebulized COPD treatment into a first-line dual bronchodilator category?
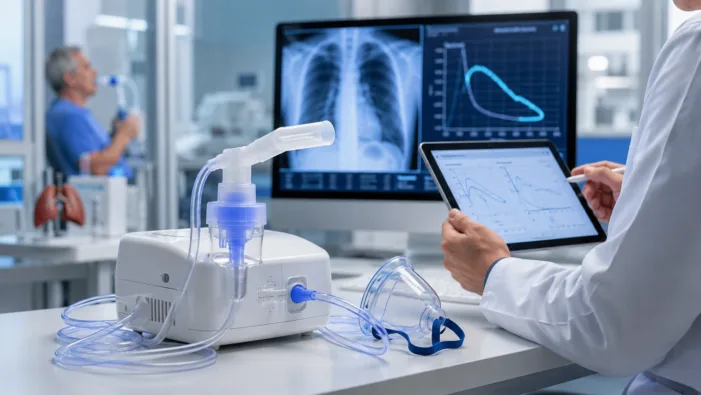
COPD care relies on handheld inhalers, but many patients struggle with delivery. AeroRx is testing whether nebulized dual therapy can close that gap.

COPD care relies on handheld inhalers, but many patients struggle with delivery. AeroRx is testing whether nebulized dual therapy can close that gap.
GSK plc has received regulatory approval from China’s National Medical Products Administration (NMPA) to market Trelegy Ellipta, a once-daily inhaled combination of fluticasone furoate, umeclidinium, and vilanterol, for the treatment of adult patients with uncontrolled asthma. This decision expands Trelegy Ellipta’s label beyond its existing indication in chronic obstructive pulmonary disease (COPD), making it the […]

AirNexis launches with $200M and ex-China rights to AN01. Find out why this dual-pathway COPD therapy is drawing investor attention in 2026.

GSK plc’s latest agreement with the United States Administration to lower the price of respiratory medicines, including its inhaled asthma and chronic obstructive pulmonary disease (COPD) portfolio, signals a shift in how global pharmaceutical companies are choosing to respond to increasing U.S. political pressure around drug affordability. The agreement, announced on December 22, 2025, satisfies […]
Mabwell Biopharmaceuticals has received Investigational New Drug (IND) clearance from the United States Food and Drug Administration to initiate a Phase IIa clinical study for its anti-ST2 monoclonal antibody, 9MW1911, in patients with moderate-to-severe chronic obstructive pulmonary disease. This regulatory milestone comes on the heels of a completed Phase IIa study in China, where Mabwell […]