Why 3Shape’s Dx Software clearance matters for digital dentistry’s next phase
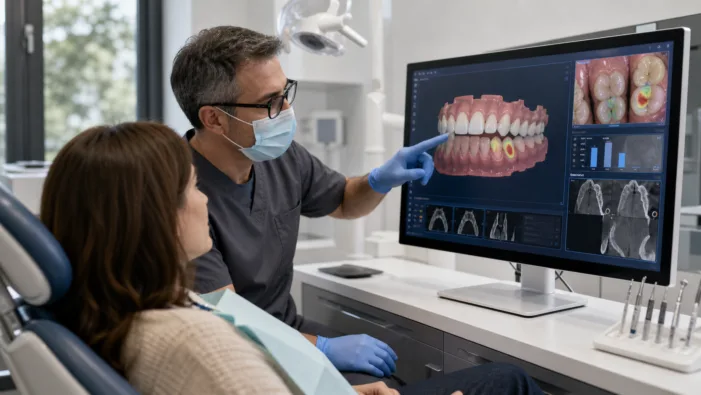
Dental AI is moving from scan capture to diagnosis support. 3Shape Dx now faces the harder test of U.S. clinician adoption.

Dental AI is moving from scan capture to diagnosis support. 3Shape Dx now faces the harder test of U.S. clinician adoption.