3Shape has received U.S. Food and Drug Administration 510(k) clearance for 3Shape Dx Software, allowing the dental software platform to be marketed in the United States for use by qualified dental professionals in adult patients. The clearance covers Dx Plus and Dx Standard, positioning the software within the growing market for AI-assisted oral health assessment, intraoral scan interpretation, and patient communication tools.
Why 3Shape Dx clearance could matter beyond another digital dentistry software launch
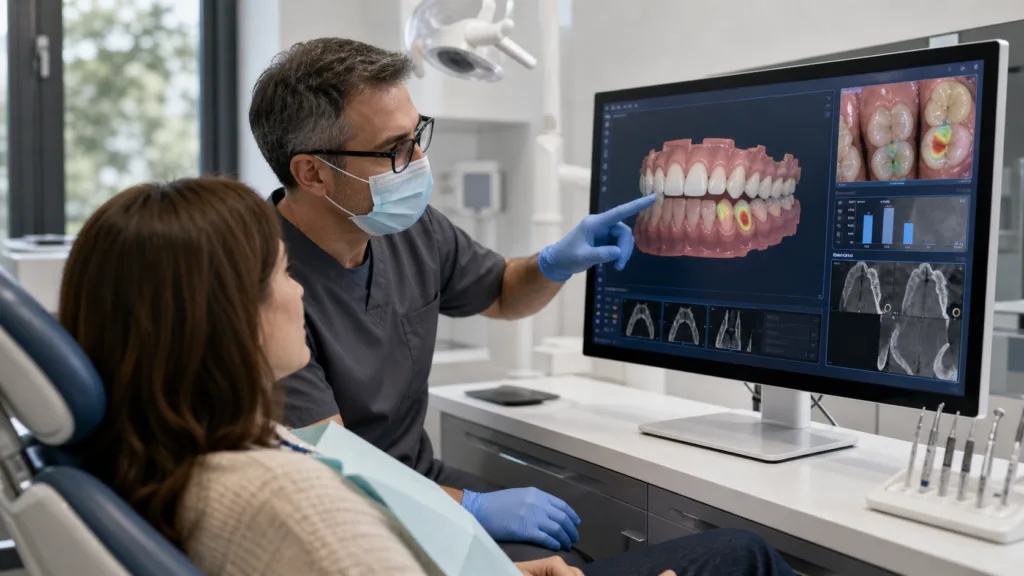
The most important signal in this clearance is not simply that another dental software tool can now enter the U.S. market. It is that intraoral scanning is being pushed further along the diagnostic value chain. For years, digital dentistry has been dominated by workflow efficiency, restorative design, clear aligner planning, implant workflows, and better digital records. 3Shape Dx Software points to a more ambitious direction, where the scan becomes not only a digital impression or visual record, but a recurring clinical reference point for detecting, tracking, and explaining oral health conditions.
That shift matters because dental practices are increasingly being asked to justify investment in digital platforms beyond speed and convenience. A scanner that supports treatment planning is valuable. A scanner-linked software layer that can help clinicians identify surface and proximal caries, plaque, tooth wear, and gingival recession may carry a different level of commercial relevance. It can touch diagnosis support, recall visits, prevention, case acceptance, and patient education, which are all central to dental practice economics.
The unresolved question is whether this moves the market toward meaningful diagnostic assistance or simply adds another visualization layer to the chairside experience. FDA clearance gives 3Shape a regulatory pathway into the United States, but adoption will depend on how dentists perceive the software in daily workflow. If the tool reduces friction, improves consistency, and helps patients understand treatment needs, it could become sticky. If it adds complexity, requires too much explanation, or produces outputs that clinicians feel they must heavily recheck, the commercial impact may be slower than the regulatory milestone suggests.
How AI-assisted dental diagnostics could change routine checkups and chairside communication
The clinical relevance of 3Shape Dx Plus sits in the routine nature of the use case. Dental practices already conduct periodic checkups, take scans, examine changes over time, and explain findings to patients who may struggle to understand what they are seeing. AI-assisted visualisation could make those conversations less dependent on abstract descriptions and more anchored in chairside images that show areas of concern in a clearer format.
That is commercially important because dentistry often depends on patient understanding and trust. A clinician may identify tooth wear or early gingival recession, but patient acceptance can be uneven when the issue does not yet feel urgent. A software tool that helps convert scan data into visual evidence may support better conversations around prevention, monitoring, and treatment planning. In that sense, 3Shape Dx is not only competing as a diagnostic aid. It is competing as a communication product.
However, the strength of this opportunity also creates a risk. Visuals can influence patient perception, and the line between education and over-interpretation must be carefully managed. Dental professionals will need to understand how the software should be used, what its limitations are, and how to explain findings without implying that the software alone has made a diagnosis. The practical test will be whether 3Shape Dx supports clinician judgement rather than crowding it with software-driven confidence that may not always match clinical nuance.
What the clearance reveals about 3Shape’s strategy in the intraoral scanner ecosystem
The compatibility structure is strategically revealing. Dx Plus is exclusively compatible with 3Shape TRIOS 6 scans, while Dx Standard is compatible with all 3Shape TRIOS models except TRIOS Core. That creates a clear ecosystem incentive. For customers already invested in 3Shape TRIOS scanners, Dx Software may increase platform value. For practices considering scanner upgrades, AI-assisted diagnostic support could become part of the decision calculus.
This is where the move becomes more than a software clearance. 3Shape appears to be reinforcing the scanner as the hub of a broader digital dentistry platform. Intraoral scanner makers are no longer competing only on scan speed, accuracy, ergonomics, or integration with labs. They are increasingly competing on what the scan enables after capture. That post-scan software layer may become a major differentiator as hardware performance matures and dental practices look for clearer return on investment.
The limitation is that ecosystem-based value can cut both ways. Exclusive compatibility with TRIOS 6 for Dx Plus may strengthen loyalty among 3Shape users, but it also narrows the immediate addressable base for the most advanced version of the software. Dental clinics with older scanner fleets or mixed digital systems may hesitate if they perceive the AI-assisted functionality as tied too tightly to a hardware upgrade cycle. For 3Shape, the commercial upside will depend on whether the software feels compelling enough to drive upgrades without making customers feel boxed in.
Why FDA 510(k) clearance gives 3Shape a clearer U.S. route but not a guaranteed adoption curve
The 510(k) clearance provides a commercial gateway into the United States, one of the most important dental technology markets globally. It also gives dentists, clinics, and dental service organizations a regulatory reference point when evaluating whether to deploy the software in practice. For a product operating near diagnosis support, that matters. Dental AI tools face a higher trust threshold than administrative software or imaging storage tools because they sit closer to clinical decision-making.
The clearance also suggests that regulators are becoming increasingly familiar with software-enabled dental tools that aid assessment rather than replace professional judgement. That distinction is central. 3Shape Dx Software is positioned for qualified dental professionals, which keeps the clinician in control and frames the technology as assistive. This may help with regulatory acceptance, professional adoption, and risk management.
Yet regulatory clearance is not the same as clinical proof in the eyes of every buyer. Dental professionals may still ask how the software performs across patient populations, scan quality variations, lesion types, gingival presentations, and real-world clinic conditions. They may also want to understand false positives, false negatives, usability in busy practices, and how outputs compare with established examination methods. Regulatory watchers may see the clearance as a meaningful step, but clinicians will judge the platform by consistency, explainability, and whether it improves decisions without slowing appointments.
What could make 3Shape Dx commercially attractive to dental service organizations
Dental service organizations may be among the more important customer groups to watch. Large clinic networks often seek standardised workflows, better documentation, consistent patient communication, and tools that can support training across multiple sites. A software platform that helps quantify and track dentition changes could be valuable in that environment, particularly if it supports repeatable chairside workflows and creates a clearer record of oral health progression.
The Dx Standard version may have a role here because manual tracking and quantification can still add value even without the AI-assisted detection features of Dx Plus. For many networks, standardisation may matter as much as automation. If teams can use scans to show changes in tooth wear, gingival recession, or other visible conditions over time, the platform could support prevention-focused dentistry and improve patient engagement across recurring visits.
The risk is that dental service organizations tend to be disciplined buyers. They will likely assess training burden, subscription costs, hardware requirements, integration with existing practice management systems, and measurable impact on chair time or treatment acceptance. A promising clinical interface is not enough if it does not fit operational workflows. For 3Shape, the enterprise opportunity may depend on showing that Dx Software can work at scale, not just impress in demonstrations.
How 3Shape Dx compares with the broader direction of dental AI and imaging software
Dental AI is already active across radiograph interpretation, charting support, treatment planning, orthodontics, implant workflows, and insurance-related documentation. 3Shape Dx Software sits in a somewhat different lane because it is linked to intraoral scan data rather than conventional two-dimensional dental radiography. That matters because scans capture visible surfaces, morphology, gingival contours, and longitudinal change in a way that can be useful for patient-facing explanations.
This does not make intraoral scan-based assessment a substitute for other diagnostic tools. Instead, it may become complementary. Radiographs remain central for many forms of detection, especially where internal or interproximal information is needed. Visual examination, periodontal probing, clinical history, and professional judgement remain essential. The strategic value of Dx Software is more likely to come from combining scan-based visualisation with the broader clinical workflow rather than displacing existing diagnostic methods.
Industry observers are likely to watch whether dental AI platforms begin to converge. If scan-based tools, radiograph AI, practice management systems, and treatment planning software remain separate, clinicians may face more screen switching and fragmented interpretation. If platforms integrate smoothly, the next wave of dental AI could become less about isolated detection features and more about unified clinical decision support. 3Shape’s advantage may lie in its scanner ecosystem, but the long-term competitive question is whether that ecosystem can connect effectively with the rest of the dental technology stack.
Why patient communication may be the most immediate use case for 3Shape Dx
Although the AI-assistive capabilities will naturally attract attention, the near-term commercial impact may come from patient communication. Dentistry has a persistent communication challenge because many oral health issues are gradual, asymptomatic, or difficult for patients to interpret. A patient may not fully grasp why tooth wear, plaque accumulation, early caries, or gingival recession matters until the condition is shown clearly and tracked over time.
Dx Plus visualisations could help clinicians explain findings more clearly, while Dx Standard could support longitudinal monitoring. That combination may improve trust when treatment recommendations are made. The value proposition is not only detection, but also clarity. In a market where patients increasingly expect visual, digital, and transparent explanations, that could be a powerful adoption driver.
However, patient communication tools must be handled carefully. Overly vivid visualisations can create anxiety or confusion if not explained properly. Clinicians will need to frame the software output as part of a professional assessment, not as a standalone verdict. The best outcome for 3Shape would be a workflow in which Dx Software strengthens the clinician-patient relationship. The weaker outcome would be a tool that patients perceive as persuasive technology rather than balanced clinical communication.
What clinicians and industry observers are likely to watch after the U.S. launch
The next phase will be less about the clearance itself and more about market evidence. Clinicians will watch whether the software is fast enough for routine checkups, whether outputs are easy to interpret, and whether it improves confidence in monitoring change over time. Practice owners will examine whether it supports productivity, case conversion, patient retention, and return on investment. Dental service organizations may assess whether it can standardise workflows across multiple locations.
For regulators and industry observers, the broader question is how AI-assisted dental software will be governed as products become more capable. Tools that aid detection and visualisation may gradually expand into more sophisticated assessment categories. That makes transparency, training, performance monitoring, and post-market feedback increasingly important. The more these tools influence clinical conversations, the more scrutiny they may attract around accuracy, bias, and appropriate use.
For 3Shape, the opportunity is clear. The diagnostics-focused dental technology developer has a chance to deepen the value of its TRIOS ecosystem and strengthen its position in digital dentistry at a time when AI-assisted tools are moving from novelty to workflow infrastructure. The challenge is equally clear. U.S. dentists will not adopt Dx Software just because it has clearance. They will adopt it if it makes examinations clearer, patient conversations easier, and clinical workflows more consistent without creating new layers of uncertainty.
3Shape Dx clearance is a platform signal, not just a product milestone
The stronger interpretation of this clearance is that 3Shape is trying to shift the role of intraoral scanning from documentation to active clinical support. That is a bigger strategic move than simply adding another software module. If Dx Software becomes embedded in routine checkups, 3Shape could strengthen the commercial moat around TRIOS scanners and make scan-based monitoring a more central part of everyday dentistry.
The more cautious interpretation is that the U.S. market will demand proof of practical value. Dental clinics are busy, cost-sensitive, and already managing multiple digital tools. AI-assisted software must be accurate, explainable, easy to use, and commercially rational. The clearance opens the door, but it does not guarantee that dentists will walk through it at scale.
The most likely near-term outcome is selective adoption among digitally mature practices, TRIOS 6 users, and clinics that already use scans heavily for patient engagement. Broader adoption may take longer and will depend on training, workflow fit, perceived accuracy, and whether the software can show value beyond impressive visuals. For 3Shape, the U.S. launch is not the finish line. It is the start of the real commercial exam.