Is Amgen’s bispecific lung cancer drug becoming a global standard after chemotherapy failure?
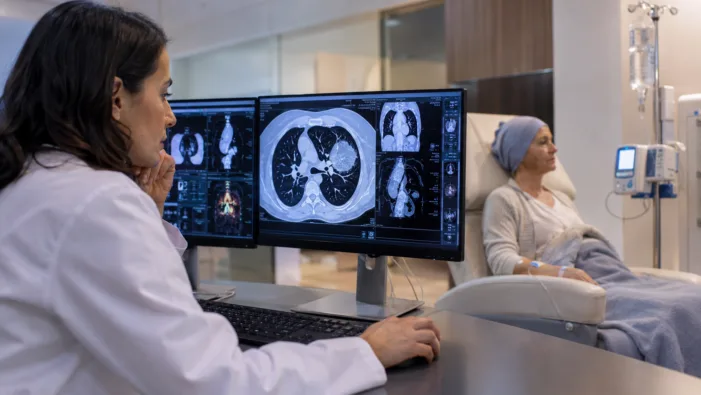
Amgen’s Imdylltra has European approval in relapsed small cell lung cancer. Survival data now face reimbursement and adoption tests.

Amgen’s Imdylltra has European approval in relapsed small cell lung cancer. Survival data now face reimbursement and adoption tests.
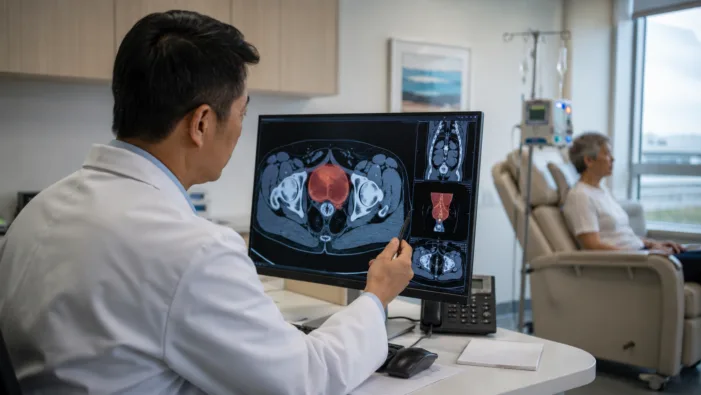
Merck’s bladder cancer push is moving earlier in Europe. KEYTRUDA plus Padcev could reset cisplatin-ineligible MIBC care.

PYRUKYND has EU approval in thalassaemia. Now Agios and Avanzanite face Europe’s harder test: access, uptake and reimbursement.

Hemophilia care is moving beyond factor replacement. Pfizer’s Hympavzi EU expansion tests how far anti-TFPI therapy can scale.
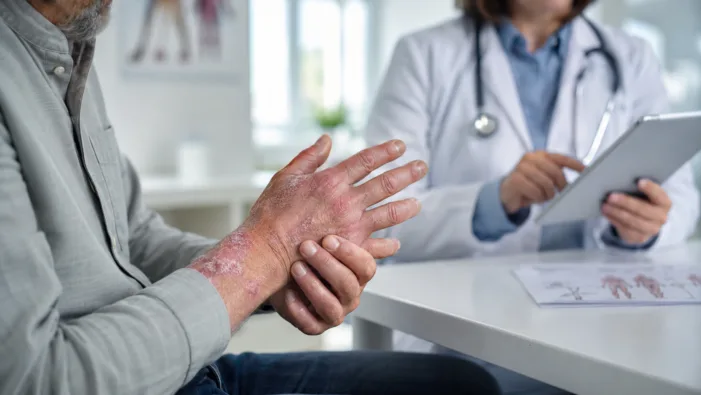
A new EU approval is only the first hurdle. Sotyktu now faces Europe’s tougher test: payer access, sequencing, and real-world uptake.

Discover how Rhythm Pharmaceuticals, Inc.’s EU approval of setmelanotide could redefine rare obesity treatment and what comes next for the market.

European Union approval gives Merck’s ENFLONSIA a new opening in infant RSV prevention. Read what it means for rollout, reimbursement, and competition.

EU orphan status for morcamilast in PPP reshapes Meiji Seika Pharma’s European strategy. See what it means for PDE4 competition and rare dermatology.

EU approves 7.2 mg Wegovy for obesity. Explore what this means for efficacy, competition and reimbursement across Europe.

The European Commission’s recent approval of €47 million in German state aid for Vetter Pharma’s new aseptic filling facility in Saarlouis marks more than a regional win. It signals a structural shift in how Europe views contract manufacturing organizations, or CDMOs—not merely as outsourcing vendors, but as core pillars of supply chain resilience, regional economic […]