Ribo’s coronary artery disease filing highlights the next frontier for RNA-based cardiovascular drugs

Ribo’s RBD1119 Phase 2 filing could test whether siRNA drugs can reduce coronary artery disease risk without bleeding trade-offs.

Ribo’s RBD1119 Phase 2 filing could test whether siRNA drugs can reduce coronary artery disease risk without bleeding trade-offs.
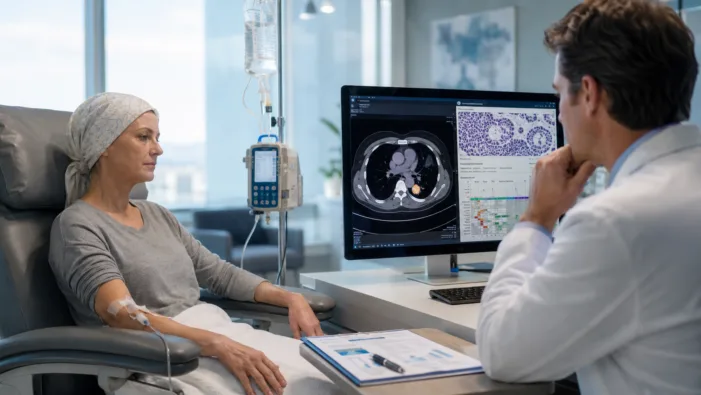
Find out how Enhertu’s EU recommendation could reshape HER2 targeted treatment for metastatic solid tumors.

Gilead’s Trodelvy may move earlier in metastatic TNBC. The EU signal could reshape first-line ADC use, but adoption questions remain.
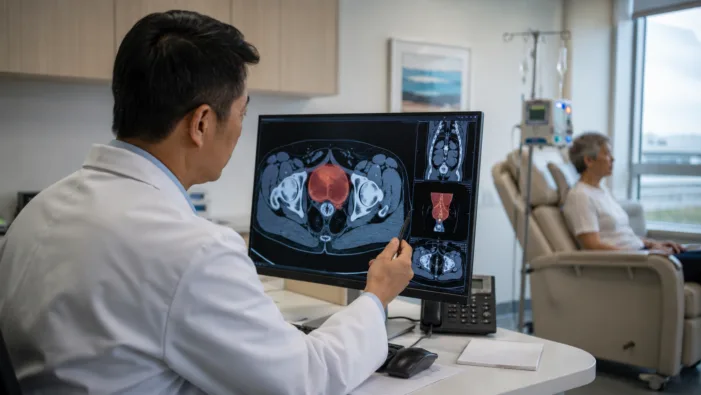
Merck’s bladder cancer push is moving earlier in Europe. KEYTRUDA plus Padcev could reset cisplatin-ineligible MIBC care.

Biomarkers can speed drug approvals, but weak validation can haunt sponsors. FDA and EMA are tightening the path from signal to endpoint.

Hemophilia care is moving beyond factor replacement. Pfizer’s Hympavzi EU expansion tests how far anti-TFPI therapy can scale.
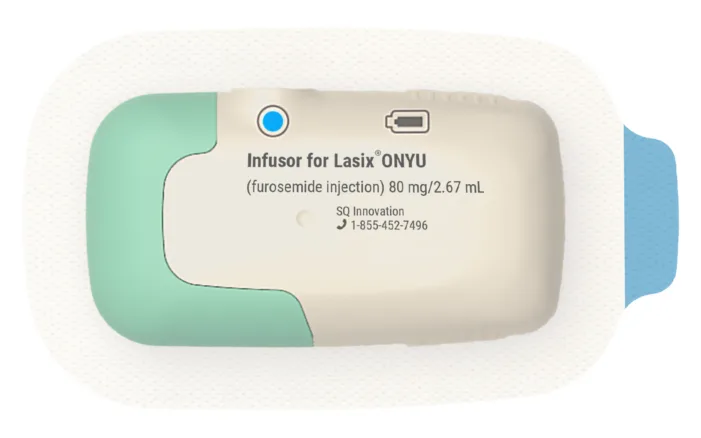
Heart failure care needs fewer hospital stays. Lasix ONYU now tests whether Europe is ready for at-home subcutaneous diuresis.

Plozasiran clears CHMP but the real test is reimbursement. Europe’s HTA bodies will now decide whether clinical FCS diagnosis is enough.

Novartis Pharma AG’s Itvisma gains CHMP support in SMA. Find out how gene therapy could reshape treatment for older patients in Europe.

Read how Novo Nordisk may be reducing one of the biggest GLP-1 delivery barriers in Europe and what it could mean for Wegovy’s market expansion.