Why clinical biomarkers face a slow climb before regulators accept them as surrogate endpoints

Biomarkers can speed drug approvals, but weak validation can haunt sponsors. FDA and EMA are tightening the path from signal to endpoint.

Biomarkers can speed drug approvals, but weak validation can haunt sponsors. FDA and EMA are tightening the path from signal to endpoint.

HyBryte’s setback leaves Soligenix with cash, orphan assets, and a harder question. Can SGX945 reset the rare disease story?

VT ablation remains difficult. Adagio Medical’s pivotal vCLAS data now shifts the question from feasibility to FDA and clinical adoption.

Savara’s molgramostim FDA review was extended to November 22, 2026. Read what this means for autoimmune PAP, launch timing, and regulatory risk.

Parnell’s Noble Pharma acquisition expands U.S. veterinary drug manufacturing capacity. Read what it could change for supply, scale, and market strategy.
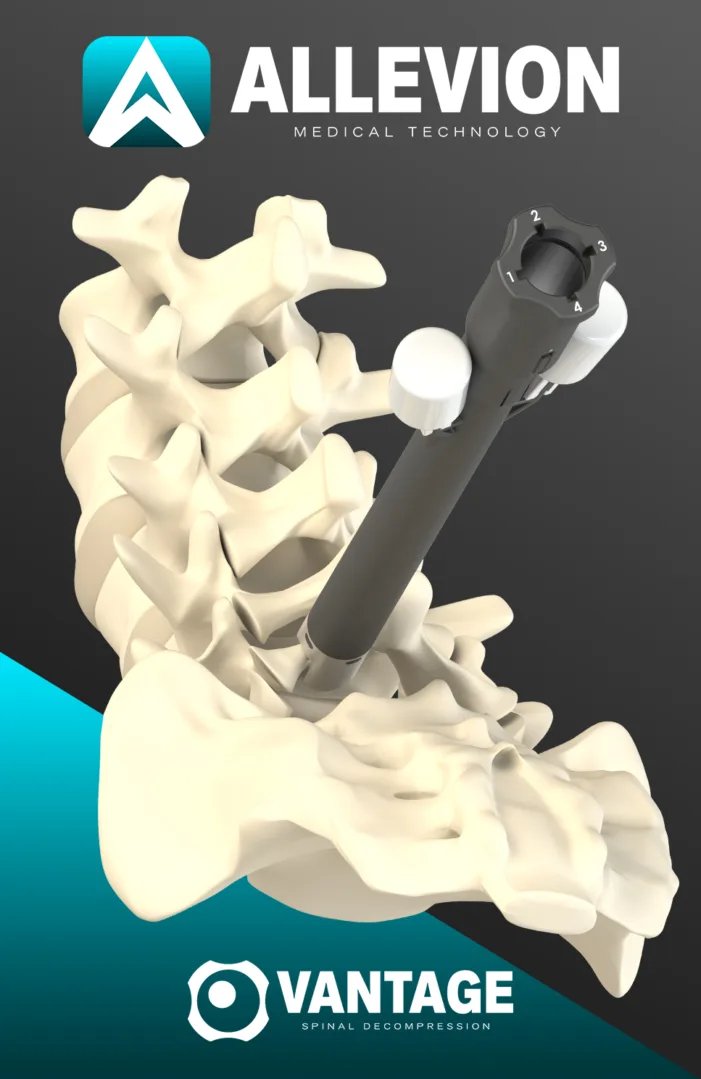
Allevion Medical’s Vantage won FDA 510(k) clearance for lumbar decompression. Read what this may change for spinal stenosis workflow, adoption, and risk.

Novo Nordisk’s Awiqli wins FDA approval as the first once-weekly basal insulin. Read what this could change for type 2 diabetes care.