Can Moderna turn hantavirus urgency into a credible rare-virus vaccine strategy?

Moderna surged on hantavirus hopes. The real test is whether rare-virus urgency can become a viable mRNA vaccine pathway.

Moderna surged on hantavirus hopes. The real test is whether rare-virus urgency can become a viable mRNA vaccine pathway.

Hantavirus has no specific antiviral option. Traws Pharma’s next test is turning outbreak urgency into credible clinical candidates.

Meiji Seika Pharma has launched Meiji Pharma Asia in Singapore. Read what this ASEAN expansion could change for vaccines and regional pharma growth.

Vedanta Biosciences kept VE303 Phase 3 on track after interim review. Read what this means for recurrent C. difficile treatment and microbiome drug development.

Find out how Bavarian Nordic’s Brazil rollout with Eurofarma puts its chikungunya vaccine to the ultimate regulatory and execution test.
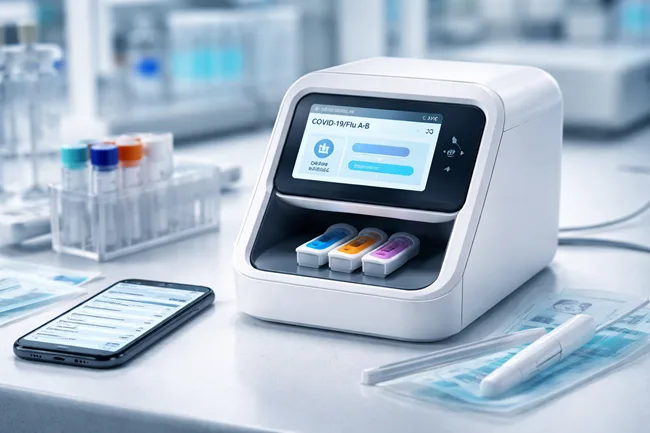
Co-Diagnostics, Inc. has received its first international patent for its Co-Dx PCR platform, with the Australian Patent Office granting coverage for the point-of-care molecular diagnostics system, including its PCR Pro instrument and disposable test cups. This development marks a critical step in the U.S.-based company’s international intellectual property strategy as it advances toward global commercialization […]

Meiji Seika Pharma Co., Ltd. has announced a strategic partnership with MBC BioLabs, a leading biotech incubator based in California’s San Francisco Bay Area, to expand its global drug discovery initiatives. The Tokyo-headquartered pharmaceutical firm aims to deepen its open innovation approach by tapping into early-stage biotech talent in the United States, with a focus […]

Innoviva Specialty Therapeutics has received approval from the U.S. Food and Drug Administration for NUZOLVENCE (zoliflodacin), a first-in-class, single-dose oral antibiotic for the treatment of uncomplicated urogenital gonorrhea in adults and adolescents. This approval, based on the largest-ever Phase 3 clinical trial for a new treatment against Neisseria gonorrhoeae, marks the first new oral gonorrhea […]