Kelun-Biotech’s ASCO 2026 data strengthens China’s next wave of targeted cancer drugs
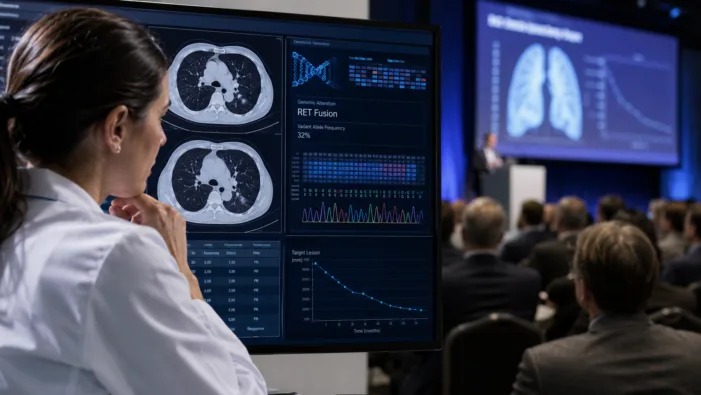
Kelun-Biotech’s RET lung cancer data looks strong, but CNS durability and global competition will decide its real impact.

Kelun-Biotech’s RET lung cancer data looks strong, but CNS durability and global competition will decide its real impact.

A TROP2 ADC just pushed past pembrolizumab alone in first-line lung cancer. See why OptiTROP-Lung05 matters now.
Ono’s ONO-2808 data hints at disease modification in MSA. See why Phase 3 may decide whether the signal becomes a breakthrough.

Leqembi’s Alzheimer’s rollout is still slow, but Eisai’s 2028 sales simulation keeps BioArctic’s royalty story alive.

Cell therapy scale-up is not just a science problem. Charles River and MEDIPOST are targeting the GMP testing bottleneck.

China has hepatitis B scale. GSK has bepirovirsen. SBP Group may decide whether science can become real-world access.
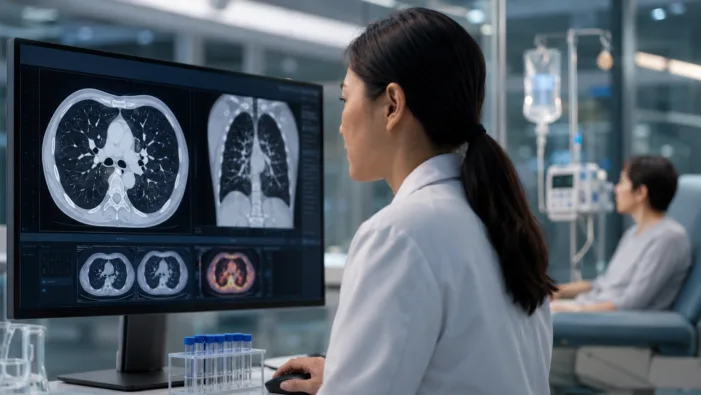
ADC ambition meets Keytruda dominance. Kelun-Biotech’s sac-TMT filing could test how far TROP2 combinations can move in lung cancer.
Alzheimer’s drugs need easier delivery. Eisai and Biogen now face an FDA timing test that could shape Leqembi’s access story.
NUT carcinoma has few options and poor survival. Wenda Pharma’s NHWD-870 may test how fast China can move rare oncology forward.
Insulin access needs more than approvals. LANGLARA’s real test is whether payers and pharmacies turn interchangeability into savings.