Kelun-Biotech’s ASCO 2026 data strengthens China’s next wave of targeted cancer drugs
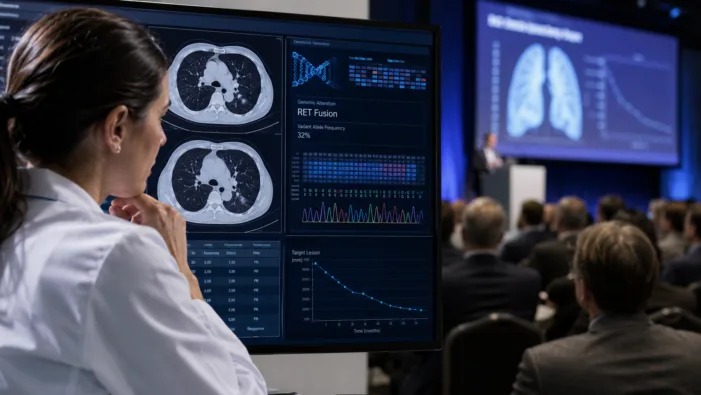
Kelun-Biotech’s RET lung cancer data looks strong, but CNS durability and global competition will decide its real impact.

Kelun-Biotech’s RET lung cancer data looks strong, but CNS durability and global competition will decide its real impact.

EP-NEC has no approved drug worldwide. Leads Biolabs now has to prove Opamtistomig can turn rare-tumor urgency into regulatory momentum.
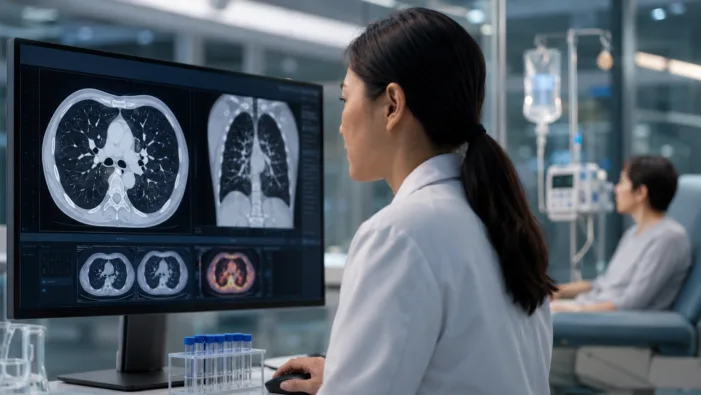
ADC ambition meets Keytruda dominance. Kelun-Biotech’s sac-TMT filing could test how far TROP2 combinations can move in lung cancer.

Lynk Pharmaceuticals won NMPA acceptance for zemprocitinib in atopic dermatitis. Read what this filing changes, and what risks still remain.
GSK plc has received regulatory approval from China’s National Medical Products Administration (NMPA) to market Trelegy Ellipta, a once-daily inhaled combination of fluticasone furoate, umeclidinium, and vilanterol, for the treatment of adult patients with uncontrolled asthma. This decision expands Trelegy Ellipta’s label beyond its existing indication in chronic obstructive pulmonary disease (COPD), making it the […]

Akeso’s gumokimab enters NMPA review for ankylosing spondylitis after Phase 3 success. Find out what this could mean for IL-17 therapies in China.