What Acupath’s ArteraAI Prostate Test rollout reveals about the next phase of precision oncology
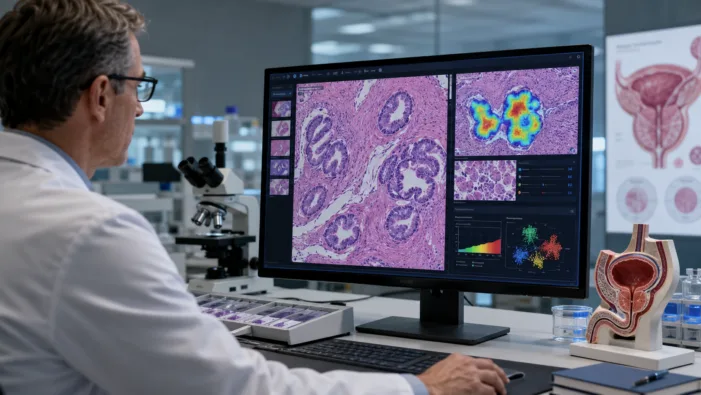
Acupath adds ArteraAI Prostate Test to its pathology workflow. Find out what it means for AI prostate cancer risk stratification today.

Acupath adds ArteraAI Prostate Test to its pathology workflow. Find out what it means for AI prostate cancer risk stratification today.

Hologic’s Breast Cancer Index is changing endocrine therapy decisions. The bigger test is adoption in real-world breast cancer care.

MRD testing is moving from research to reimbursement. Veracyte now has Medicare coverage, but adoption in bladder cancer care is the harder test.
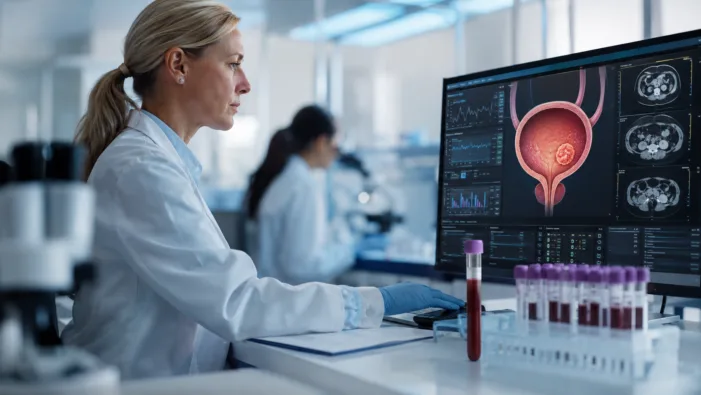
MRD testing has payer momentum, but clinical adoption is tougher. Veracyte’s TrueMRD Medicare win puts bladder cancer monitoring in focus.
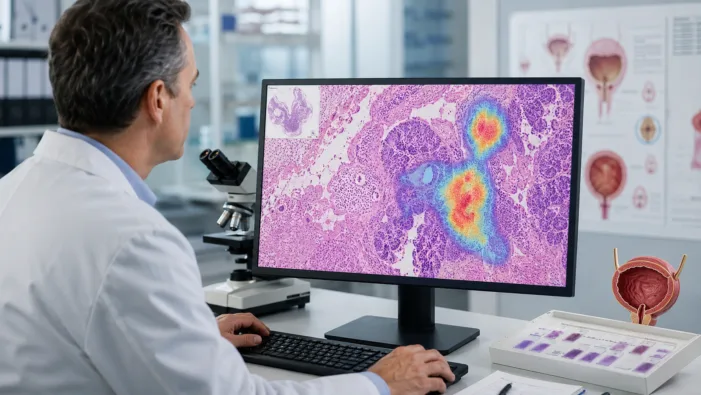
Bladder cancer risk tools leave gray zones. Valar Labs’ FDA breakthrough nod tests whether AI pathology can narrow them.
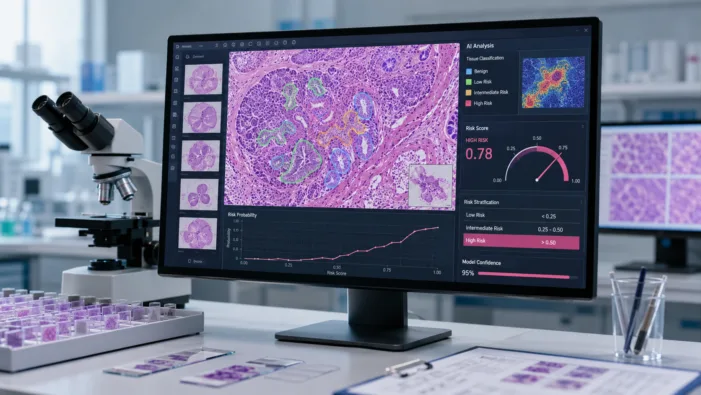
AI can read pathology slides, but can it change breast cancer treatment decisions? ArteraAI Breast puts that question before clinicians and payers.

Roche wants AI pathology at scale. PathAI gives it the tissue-analysis layer needed to tighten diagnostics, trials, and precision oncology.

Plus Therapeutics has won an AMA PLA code for CNSide. Read why this reimbursement milestone could shape adoption, coverage, and diagnostic growth.

Junction and GRAIL are expanding Galleri access, but can workflow integration overcome regulatory, reimbursement, and adoption hurdles? Read more.

Radiopharm Theranostics reports strong RAD 101 interim data in brain metastases imaging. Read expert analysis on clinical impact and regulatory outlook.