Why Servier’s FIREFLY-2 enrollment milestone could reshape pediatric glioma treatment
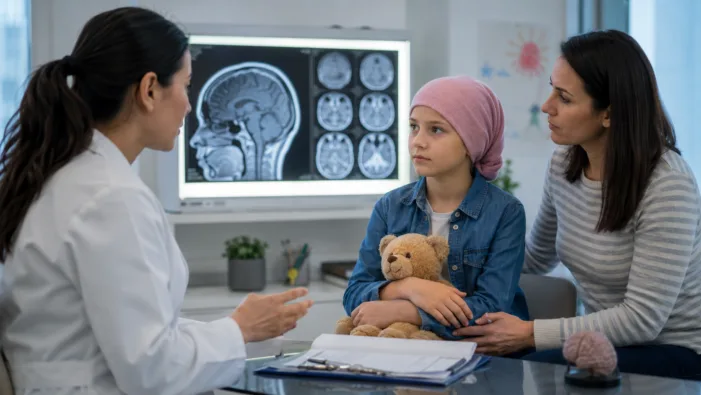
OJEMDA is already approved after relapse. FIREFLY-2 asks whether Servier can move targeted pLGG therapy earlier.

OJEMDA is already approved after relapse. FIREFLY-2 asks whether Servier can move targeted pLGG therapy earlier.
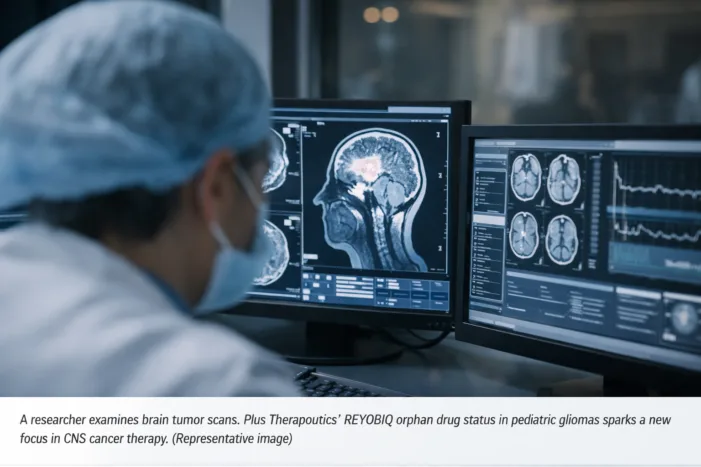
Plus Therapeutics won orphan drug designation for REYOBIQ in pediatric malignant gliomas. Read what it changes for CNS cancer development.

Ipsen nears EU approval for Ojemda in BRAF-driven pediatric glioma. Explore regulatory impact, clinical implications, and long-term risks.

Theriva’s APAO 2026 VCN-01 data reveals how viral synergy and local delivery could redefine pediatric eye cancer care.
FDA approves YARTEMLEA for TA-TMA. Find out how Omeros’ new drug could change transplant care for children and adults alike.

Orphalan, the international pharmaceutical company known for developing orphan drugs, has announced the acquisition of Orphelia Pharma, a specialist in rare and severe pediatric diseases. The deal enhances Orphalan’s position in the European rare disease market, particularly within pediatric neurology and oncology, and signals a strategic pivot toward deeper vertical integration in pediatric formulations and […]