Carl Zeiss Meditec AG confirmed on February 2, 2026, that it has received National Medical Products Administration (NMPA) approval in China for its ARTEVO 750 and ARTEVO 850 ophthalmic surgical microscopes. The regulatory clearance allows ZEISS to offer its most advanced digital and optical visualization platforms to Chinese surgeons performing cataract, retinal, glaucoma, and corneal procedures. Both microscope systems are now cleared for commercial deployment in one of the world’s fastest-growing ophthalmic device markets.
What this approval signals about the trajectory of surgical visualization in Chinese ophthalmology
China’s approval of the ARTEVO 750 and 850 surgical microscopes reflects more than just a regulatory milestone for ZEISS. It signals a broader inflection point in the digitalization of surgical infrastructure across China’s ophthalmic centers. The NMPA’s greenlight suggests that complex systems blending optical performance, digital overlays, workflow software, and data interoperability are increasingly aligned with Chinese healthcare priorities.
The ARTEVO 850, with its 3D digital visualization capabilities and modular integration with ZEISS’s CALLISTO eye surgical planning platform, is designed not only to improve image clarity but also to reduce intraoperative fatigue, enhance anatomical precision, and enable surgeon-driven customization. For institutions juggling high volumes of cataract and retina surgeries—especially in tertiary academic hospitals—the shift to digitally orchestrated surgical environments promises increased throughput with fewer complications.
Meanwhile, the ARTEVO 750, which sits lower in the product hierarchy but incorporates intraoperative OCT, RGB-enhanced illumination, and a deepened depth of field, serves as an intermediary platform. It targets surgeons transitioning from analog to digital while preserving traditional ZEISS optical quality.
Industry analysts familiar with the Chinese market see this dual-platform approach as an astute segmentation strategy. The ARTEVO 850 caters to fully digital, high-acuity surgical teams, while the ARTEVO 750 offers a bridge for legacy institutions not yet ready for full 3D adoption but eager to integrate intraoperative imaging and enhanced visualization into routine workflows.
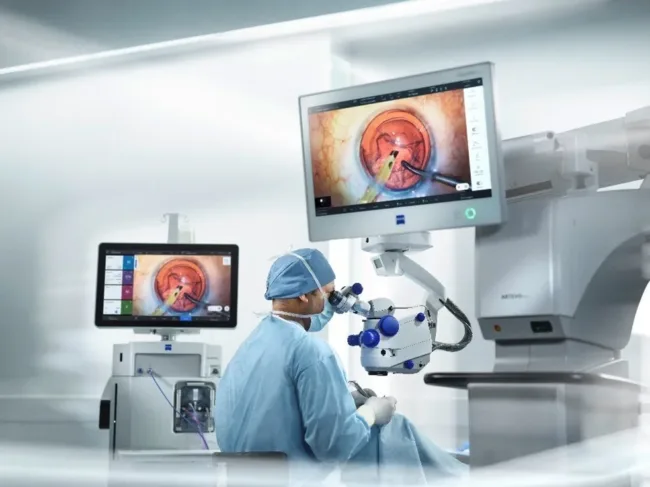
What sets ARTEVO 850 apart from competing ophthalmic microscope platforms
While digital ophthalmic microscopes have existed for over a decade, few have achieved broad surgical adoption beyond specialized research or flagship hospital environments. ZEISS is attempting to change that with the ARTEVO 850 by embedding its system in a broader ecosystem of surgical tools, imaging software, and reference datasets.
One of its most differentiating features is Smart Depth of Field (DoF), a software-driven function that reportedly enhances image clarity by up to 60 percent compared to the prior ARTEVO 800 platform. This feature automatically maintains focus on relevant anatomical layers, reducing the need for manual adjustments and allowing surgeons to maintain situational awareness during complex intraocular maneuvers.
The platform also includes the Digital Color Assistant (DCA), which dynamically adjusts color rendering to highlight surgical planes, blood vessels, and tissue boundaries. Such digital color tuning becomes especially valuable in vitrectomies or corneal transplants where visibility margins are narrow and lighting angles shift frequently.
Beyond visualization, ZEISS has pushed ARTEVO 850’s integration into a full workflow system. The microscope communicates with ZEISS CALLISTO eye for toric IOL alignment, allows reference image matching on-screen, and supports data logging—all of which enable a more unified surgical record and post-op review.
Comparatively, platforms from competitors such as Leica Microsystems and Topcon do offer 3D digital features, but they tend to lack the same degree of intraoperative software orchestration or embedded workflow support. Alcon’s NGENUITY 3D Visualization System, while competitive in certain retina settings, is not as broadly integrated with planning platforms or OCT data streams.
What this reveals about ZEISS’s commercial strategy in China’s medtech ecosystem
ZEISS’s decision to pursue a dual launch of ARTEVO 750 and 850 within China suggests a layered commercial approach designed to accelerate uptake while reducing channel friction. While premium platforms like ARTEVO 850 will likely be marketed to tier-1 hospitals and surgical training institutions, the 750’s more accessible price point and mixed analog-digital capabilities could help ZEISS penetrate tier-2 and tier-3 hospital segments over time.
The rollout also aligns with ZEISS’s increased visibility at Chinese trade shows and government-partnered medical events. The 2025 unveiling of ARTEVO 750 at the China International Import Expo (CIIE) signaled strategic alignment with state-sponsored innovation channels. NMPA approval now unlocks commercial potential—but future success may hinge on ZEISS’s ability to align with regional distributor networks, localized training programs, and potentially domestic manufacturing partnerships that would meet China’s medtech localization guidelines.
Medical device procurement in China is increasingly centralized and cost-pressured, with government programs pushing for high-tech imports to prove their comparative value or risk substitution by domestic alternatives. ZEISS’s ability to position ARTEVO as a digitally differentiated, workflow-centric platform—rather than just a microscope—may give it an edge in justifying premium placement within public tender systems.
What remains unresolved around clinical integration, training, and AI-readiness
While the devices are now formally approved, the journey from regulatory clearance to routine clinical use involves a series of unresolved challenges. First among them is surgeon training. Unlike traditional microscopes, digital 3D visualization platforms require a recalibration of depth perception, hand-eye coordination, and user interface fluency. In multi-surgeon teams, variability in adaptation speed could slow adoption or lead to inconsistent surgical performance unless ZEISS delivers robust on-site training.
Second, hospitals must assess whether their existing operating rooms have the spatial layout and connectivity required to support fully integrated platforms. 3D visualization systems often require display monitors, head-mounted or glasses-free viewing hardware, and real-time data streaming infrastructure. Institutions will need to consider both capital expenditure and downtime for infrastructure upgrades.
Third, questions remain about the scalability of ARTEVO’s AI potential. As digital surgical platforms become more data-aware, the next frontier involves real-time intraoperative decision support, anomaly detection, and even robotic-guided microsurgery. ZEISS has yet to clearly articulate whether the ARTEVO platform is equipped with edge compute capabilities or cloud-ready interfaces for AI model integration.
Clinicians watching this space believe that the utility of digital surgical platforms will hinge not only on visualization improvements but also on how data captured during surgery is analyzed and reintegrated into postoperative care pathways. Without that loop, the full promise of surgical digitalization may remain underrealized.
What China’s approval tells us about the NMPA’s evolving stance on digital surgery platforms
The NMPA’s approval of the ARTEVO 750 and 850 reflects a growing sophistication in regulatory reviews of hybrid medical platforms. These are no longer simple Class II devices with static hardware specifications but dynamically integrated systems that combine optics, software, imaging data, and intraoperative logic. The NMPA’s capacity to assess these systems for safety, efficacy, and interoperability marks a shift toward digital-first medtech regulation.
This also raises the bar for future digital surgical entrants into China. Regulatory watchers suggest that companies bringing digital platforms into China’s operating rooms must now prepare for layered scrutiny, including cybersecurity safeguards, user interface localization, and compatibility with China’s hospital information systems.
For ZEISS, this approval places it ahead of several global competitors still awaiting clearance for their advanced digital microscopy platforms. However, the window may be narrow. Domestic players in China’s medtech sector are increasingly investing in surgical robotics, digital endoscopy, and AI-guided imaging systems. Should they achieve competitive parity in digital ophthalmology, ZEISS will need to rely on continuous innovation, localization, and ecosystem partnerships to maintain leadership.
Looking ahead: Can ZEISS turn ARTEVO into the cornerstone of a digital surgical standard?
If ZEISS can achieve critical mass adoption of ARTEVO platforms in China, it may redefine the standard surgical experience in ophthalmology—not just through visualization, but through process orchestration. By combining intraoperative imaging, surgical planning tools, and digital guidance systems, the ARTEVO 850 in particular has the potential to act as a surgical control tower. Whether this becomes the dominant model will depend on ZEISS’s ability to drive training, cost efficiency, and post-op data reintegration across Chinese hospitals.
The next wave of innovation will likely center around surgical intelligence. If ZEISS builds analytics, predictive modeling, and interoperable interfaces into future ARTEVO iterations, it could position itself not just as an imaging innovator but as a platform orchestrator across ophthalmology.
For now, NMPA approval is a strong signal—but it is only the opening gambit in what may become a high-stakes digital arms race in China’s surgical theater.