Allevion Medical has received United States Food and Drug Administration 510(k) clearance for Vantage, a fully disposable sterile system for minimally invasive lumbar decompression that the Boca Raton-based medical device developer says is designed to treat pain associated with hypertrophic bone growth in lumbar spinal stenosis. The company is positioning the platform around a locate, dilate, decompress workflow, with integrated illumination and depth-controlled bone resection as key product differentiators.
Why Allevion Medical’s Vantage clearance matters beyond another routine spinal device approval
What matters now is not simply that another spinal tool kit cleared the 510(k) pathway, but what kind of problem Allevion Medical is trying to solve. The company is not arguing that decompression itself is new. Surgical and minimally invasive lumbar decompression have long been established categories. The more specific pitch is that hospitals and ambulatory surgery centers still face friction around tray complexity, sterile processing, setup burden, and procedural consistency, and that a single-use kit could reduce some of that operational drag. That is a workflow argument as much as a clinical one, and in medical technology, workflow arguments often move faster than pure technical novelty when administrators and surgeons are both under pressure to improve efficiency.
The regulatory framing is also important. A 510(k) clearance means the Food and Drug Administration found substantial equivalence to a legally marketed predicate device, not that it independently established brand-new clinical superiority. That distinction matters because companies often present 510(k) events as major validation moments, while surgeons, procurement teams, and payers tend to ask a more practical question: does the cleared product materially improve outcomes, efficiency, or economics enough to justify changing practice.
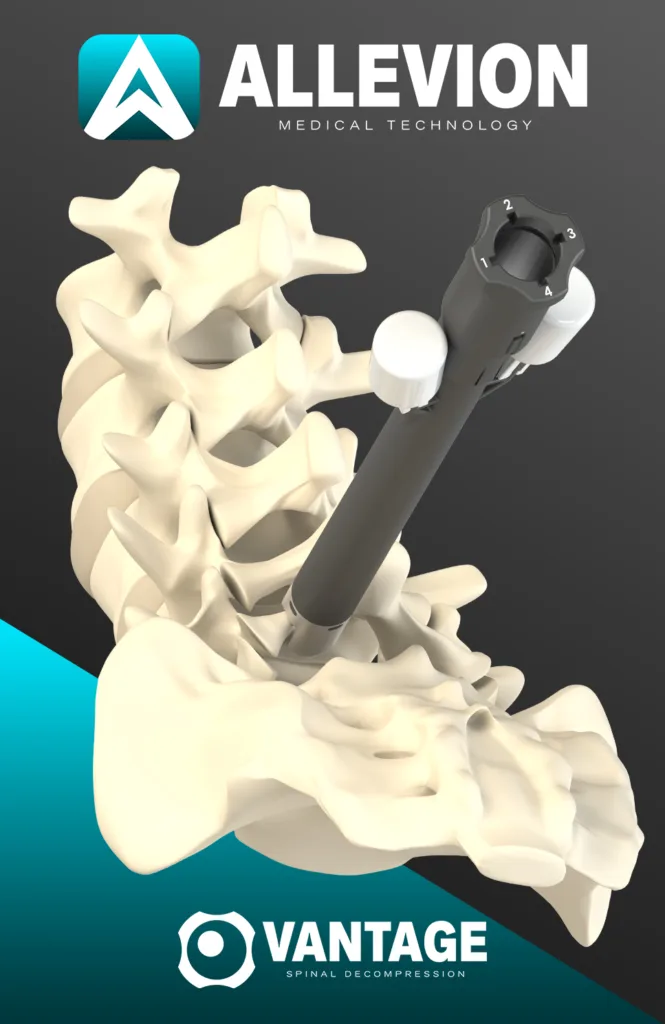
Why a fully disposable spinal decompression kit could shift operating room economics more than clinical positioning
The strongest near-term case for Vantage may be operational rather than therapeutic. Allevion Medical says the system is fully disposable and requires no additional instruments or sterile processing. In practical terms, that speaks directly to turnover time, reprocessing labor, contamination-control concerns, and the ongoing push by surgery centers to simplify inventory and reduce dependence on large reusable sets. For facilities already moving toward single-use instrumentation in selected procedures, Vantage fits a broader medtech trend in which convenience, sterilization avoidance, and logistical predictability become part of the value proposition.
That said, disposable design is not automatically an economic win. A reusable system can look expensive up front but efficient over time if case volume is high and reprocessing is well managed. A single-use system can reduce hidden labor and sterilization costs, yet still face resistance if the per-case price is viewed as too high or if administrators see little hard evidence of measurable throughput gains. This is where Allevion Medical will likely need to move beyond product language and into real-world health economic data. Industry observers tracking ambulatory spine care will likely want to see whether the company can show lower total procedural cost, shorter setup, or fewer delays linked to tray management rather than simply asserting those benefits.
How direct visualization and bone-focused decompression may shape Vantage’s clinical positioning in lumbar stenosis
The integrated illumination feature is another notable part of the pitch. Allevion Medical says Vantage offers direct visualization support through single-use integrated illumination, which it presents as a differentiator against image-guided decompression approaches that rely more heavily on indirect visualization. On paper, direct visualization may strengthen the argument for procedural control, especially when hypertrophic bone rather than ligament alone is the key target.
This is where the competitive nuance starts to matter. The minimally invasive lumbar decompression field already includes image-guided decompression approaches that are commonly described as targeting hypertrophied ligamentum flavum, with some sculpting near the lamina through a small portal. Allevion Medical is attempting to distinguish Vantage by emphasizing hypertrophic bone removal from the lamina and facet joints, while also stressing direct visualization and protective guards. That does not make the category brand new, but it does position the product differently within the decompression toolkit landscape.
That distinction could be meaningful because lumbar spinal stenosis is not a one-cause disease. Canal narrowing may involve ligamentum flavum thickening, bony overgrowth, facet arthropathy, disc-related changes, or some combination of those factors. A device designed around bone-focused decompression may appeal in cases where soft-tissue-only approaches are seen as incomplete. But this is also where the evidence bar rises. Clinicians do not just need to know what tissue can be removed. They need to know which patient profiles benefit most, what complication patterns look like, whether visualization materially changes safety, and whether the procedure preserves enough structural stability while delivering durable symptom relief.
What Vantage reveals about the difference between minimally invasive promise and device-specific proof
Comparisons with open surgery also need to be handled carefully. The company contrasts Vantage with larger-incision decompression techniques that may involve more tissue disruption and longer recovery. Broadly speaking, minimally invasive lumbar decompression has often been associated with shorter hospital stays, reduced postoperative pain burden, and lower tissue disruption relative to open laminectomy in selected patients. But that should not be confused with proof that every minimally invasive platform delivers the same benefit profile. Device-specific claims still require device-specific evidence, and platform design alone does not settle the question.
This is the central line Allevion Medical will need to walk as it commercializes Vantage. The system appears to offer a thoughtful combination of access, illumination, workflow structure, and single-use packaging. Yet surgeons and evaluators will want proof that those design choices lead to better procedural reliability or more durable patient benefit. Without that layer, the platform risks being seen as technically interesting but not sufficiently differentiated where it matters most.
Why reimbursement, training, and supply chain execution may determine whether Vantage gains real traction
Reimbursement is another major variable. Medicare coverage precedent for decompression-based approaches has helped legitimize this segment of the market, but coverage precedent for one procedural model does not automatically guarantee frictionless reimbursement for every new system entering the space. Regulatory watchers will be looking for how Vantage is coded, documented, and positioned relative to existing decompression pathways, and whether physicians view it as an incremental instrument option or a more distinct procedural platform. If coding or payer interpretation becomes messy, adoption can slow even when surgeons are intrigued by the technology.
Training will matter just as much. Allevion Medical’s locate, dilate, decompress framework is clearly designed to signal reproducibility, but spine surgeons and interventional specialists are often skeptical of simplified workflow narratives unless those claims hold up under varied anatomy and real-world complexity. Direct visualization and depth-controlled bone resection sound attractive, yet they also imply a learning curve around precision, ergonomics, and case selection. A structured workflow can reduce variability, but only if it is intuitive enough to survive outside tightly controlled early-use settings. Otherwise, the product risks being viewed as clever packaging rather than a durable change in practice.
Manufacturing and supply chain execution should not be overlooked either. Single-use systems shift some burden away from the hospital and toward the manufacturer. That means Allevion Medical will need reliable production, strong field support, dependable inventory availability, and pricing discipline if it wants surgery centers to trust a disposable-only model. In medtech, a single-use strategy is only as strong as the supply chain behind it. Missed shipments or cost inflation can quickly turn a supposed efficiency story into a procurement headache. This is especially relevant if the company hopes to scale in both hospitals and ambulatory settings, where case scheduling and inventory confidence are tightly linked.
What clinicians, regulators, and industry observers are likely to watch next after the Vantage clearance
The biggest unresolved question is clinical depth. The announcement establishes clearance and outlines design features, but it does not yet offer the kind of peer-reviewed outcomes package that would settle debates around durability, complication rates, reintervention patterns, or superiority versus incumbent minimally invasive options. Industry observers will likely watch for registry data, surgeon experience reports, and any comparative studies that test whether direct visualization plus bone-focused resection translates into better function, better pain relief, or lower downstream intervention rates. Until then, Vantage looks commercially interesting and procedurally differentiated, but still early in its proof cycle.
Seen in that light, Allevion Medical’s clearance is less a final verdict than an opening move. The Vantage system enters a lumbar stenosis market where the clinical need is well recognized, minimally invasive options already exist, and reimbursement precedent has lowered at least some barriers for decompression-based approaches. Its potential edge lies in combining disposable logistics, direct visualization support, and bone-targeted decompression in one sterile kit. Whether that becomes a genuine category shift will depend on evidence, economics, and surgeon conviction. In spine, clearance gets a company onto the field. Adoption still has to be earned case by case.